SYNTHESIS, CHARACTERIZATION AND FILM PREPARATION OF NEW CO-POLYIMIDE BASED ON NEW 3,5-DIAMINO-N-(PYRIDIN-4-YLMETHYL)BENZAMIDE, ODA AND 6FDA
- aromatic co-polyimides,
- pendant amide,
- pyridinyl pendant group
Copyright (c) 2019 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
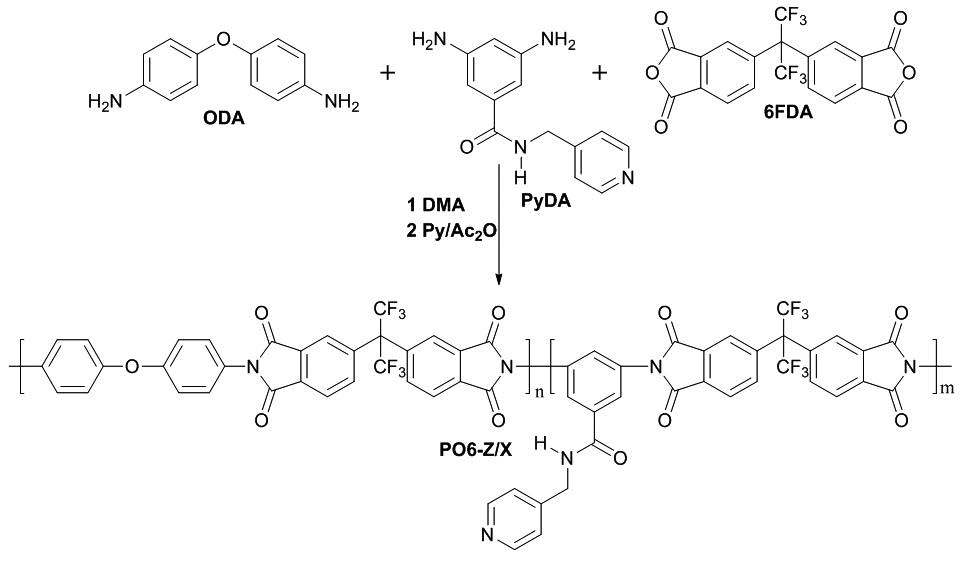
This work describes mainly the synthesis and characterization of new co-polyimides obtained from the polycondensation of the dianhydride 4,4’-(hexafluoroisopropylidene)diphthalic anhydride (6FDA), 4,4’-oxydianiline (ODA) and the new diamine 3,5-diamino-N-(pyridin-4-ylmethyl)benzamide (PyDA). It describes as the different compositions of ODA and PyDA present in the polymers, produce variation on thermal and mechanical properties, which are important characteristics for the development of future nanocomposites derived from these polymers. Both PyDA monomer and polymers were characterized using FT-IR and NMR (1H, 13C, 19F, dept 135°, COSY, HMBC, HMQC) spectroscopy. The inherent viscosity of polymers is between 0.3 to 1.49 dL/g, they are soluble in aprotic polar solvents, such as: DMSO, DMF and DMA. In addition, all co-polymers showed thermal decomposition temperature and glass transition temperatures above 483 °C and 289 °C, respectively. Mechanical tests under tension of co-polymer films showed Young’s modulus between 2.1-3.9 GPa and tensile strength between 35.5 and 120.0 MPa. On the other hand, an increase in crystallinity and hydrophilicity is observed when increasing the amount of pyridinyl groups.
References
- C.E. Sroog, J. Polymer Sci. Macromol. Rev., 11, 161 (1976).
- J. García, F. García, F. Serna y J. de la Peña, Prog Polym Sci, 35, 623, (2010).
- A.S. Dhindsa, Y.P. Song, J.P. Badyal, M.R. Bryce, Y.M. L’Vov, M.C. Petty, J. Yarwood, Chem. Mater., 4, 724 (1992).
- K. Takatoh, M. Hasegawa, M. Koden, N. Itoh, R. Hasegawa, M. Sakamoto. Alignment technologies and applications of liquid crystal devices. London, U.K.; Taylor & Francis; 2005. p. 59–63.
- D. Guzmán-Lucero, J. F. Palomeque-Santiago, C. Camacho-Zuñiga, F. Ruiz-Traviño, J. Guzmán, A. Galicia-Aguilar, C. Aguilar-Lugo, Mater., 8, 1951 (2015).
- O.J. Dautel, G. Wantz, D. Flot, J.P. Lere-Porte, J.J.E. Moreau, J.P. Parneix, F. Serein-Spirau, L. Vignau, J. Mater. Chem., 15, 4446 (2005).
- Y. Kim, J.G. Lee, K. Han, H.K. Hwang, D.K. Choi, Y.Y. Jun, J.H. Keum, S. Kim, S.S. Park, W.B. Im, Thin Solid Films, 363, 263 (2000).
- B. Smitha, S. Sridhar, A.A. Khan, J. Membra. Sci., 259, 10 (2000).
- J. Fang, X. Guo, S. Harada, T. Watari, K. Tanaka, H. Kita, K.I. Okamoto, Macromol., 35, 9022 (2002).
- M. Shigeta, M. Komatsu, N. Nakashima, Chem. Phys. Lett., 418, 115 (2006).
- K.L. Wang, Y.L. Liu, J.W., Lee, K.G. Neoh, Macromol., 43, 7159 (2010).
- J. A. Reglero, M. Trigo-López, F.C. García, J. M. García, Polymers, 9, 414 (2017).
- J.M. Gohil, P. Ray, Sep Purif Technol, 181, 159 (2017).
- M. Kim, M. Kim, B. Park, S. Kim, Desalin Water Treat, 54, 923 (2015).
- B.V. Kotov, T.A. Gordina, V.S. Voishchev, O.V. Kolninov, A.N. Pravednikov, Polym. Sci. U.S.S.R., 9, 711 (1977).
- F. Ke, N. Song, D. Liang, H. Xu, J. Appl. Polym. Sci., 127, 797 (2013).
- M. G. Dhara, S. Banerjee, Prog. Polym. Sci., 35, 1022 (2010).
- D.J. Liaw, B.Y. Liaw, C.W. Yu, Polymers, 42, 5175 (2001).
- K. Xie, J.G. Liu, H.W. Zhou, S.Y. Zhang, M.H. He, S.Y. Yang, Polymers, 42, 7267 (2001).
- C.L. Chung, W.F. Lee, C.H. Lin, S.H. Hsiao, J. Polym. Sci.: Part A: Polym. Chem., 47, 1756 (2009).
- J. M. García, F.C. García, F. Serna, J. Polym. Sci.: Part A: Polym. Chem., 41, 1202 (2003).
- C.P. Yang, R.S. Chen, K.S. Hung, Polymers, 42, 4569 (2001).
- J. Yao, C. Wang, C. Tian, X. Zhao, H. Zhou, D. Wang, C. Chen, Des. Monomers Polym., 20, 449 (2017).
- J. Zang, Z. Zhu, H. Sun, W. Liang, A. L, React. Funct. Polym., 99, 95 (2016).
- S. Zhang, X. Li, X. Guan, Y. Shi, K. Wu, L. Liang, J. Shi, M. Lu, Compos. Sci. Technol., 152, 165 (2017).
- J. Yao, C. Wang, X. Zhao, H. Zhou, C. Chen, D. Wang, High Perform. Polym., 30, 418 (2018).
- D. Liaw, K. Wang, F. Chang, Macromol., 40, 3568 (2007).
- Z. Yang, H. Wang, G. Ji, X. Yu, Yu Chen, X. Liu, C. Wua, Z. Liu, New J. Chem., 41, 2869 (2017).
- L.C. Lee, J. He, J.Q. Yu, C.W. Jones, ACS Catal., 6, 5245 (2016).
- H. Cui, Y. Chen, L. Li, Y. Wu, Z. Tang, H. Fu, Z. Tian, Microchim. Acta, 181, 1529 (2014).
- S. Pereira, A. Barros-Timmons, T. Trindade, Polymers, 10, 189 (2018).
- A. Arizaga, G. Ibarz, R. Piñol, A. Urtizberea, J. Exp. Nanosci., 9, 561 (2014).
- S. Bong, H. Yeo, B. Ku, M. Goh, N. You, Macromol Res, 26, 85 (2018).
- Z. Huang, J. Zhao, RSC Adv, 6, 34825 (2016).
- M. Barikani, S. Mehdipour–Ataei, J. Polym. Sci.: Part A: Polym. Chem., 38, 1487 (2000).
- Owens, D.K.; Wendt, R.C., J. Appl. Polym. Sci., 13, 1741 (1969).
- D. H. Kaelble, J. Adhes., 2, 66 (1970).
- J. Park, L. Drahushuk, M. Ham, S. Kang, J. Baik, S. Shimizu, M. S. Strano, C. Song, Polym Chem, 4, 290 (2013).
- N. Encinas, M. Pantoja, J. Abenojar, M. A. Martínez, J. Adhes. Sci Technol., 24, 1869 (2010).
- T.G. Woo, I.S. Park, K.H. Jung, W.Y. Jeon, K.W. Seo, Met. Mater. Int., 17, 789 (2011).
- C. R. Gautreaux, J. R. Pratt, T.L. St. Clair, J. Polym. Sci.: Part B: Polym. Phys., 30, 71 (1992).
- Y. Wang, Z. Liu, B. Han, Z. Sun, J. Zhang, D. Sun, Adv. Funct. Mater., 15, 655 (2005).


