SPECTROPHOTOMETRIC DETERMINATION OF FOUR NAPHTHALENE SULFONATES IN SEAWATER AFTER THEIR MOLECULARLY IMPRINTED STIR BAR SORPTIVE EXTRACTION
- Naphthalene sulfonates,
- molecularly imprinted polymer coated stir bar (MIPSB),
- Seawater,
- Chabahar Bay
Copyright (c) 2018 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
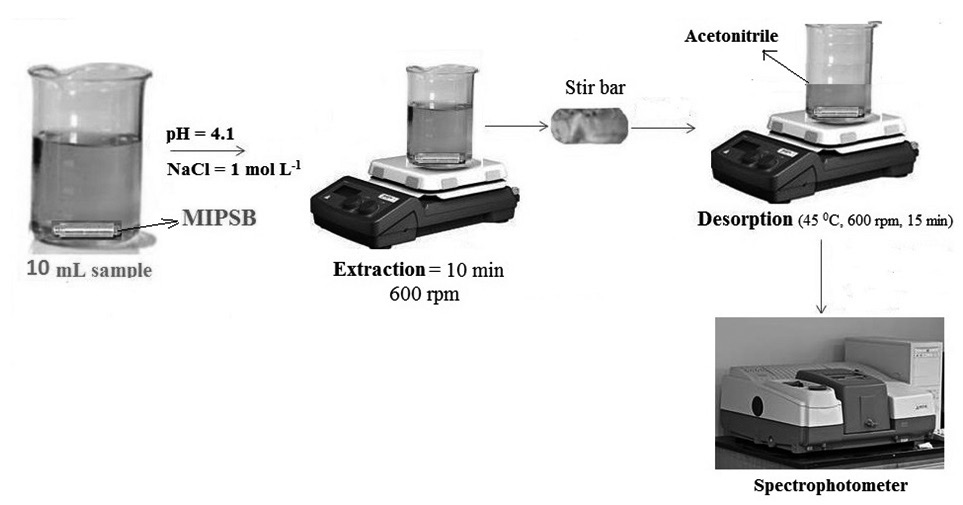
A novel 1- naphthalene sulfonic acid (1-NS) molecularly imprinted polymer (MIP)-coated stir bar for sorptive extraction of four naphthalene sulfonates (NSs) from seawater samples was prepared. 1-NS was applied as template molecule. The extraction conditions, including pH, the absorption and desorption times, stirring speed, amount of NaCl and temperature were optimized and the optimum conditions were established as: the absorption and desorption times were fixed respectively at 10 and 15 min, stirring speed was 600 rpm, pH was adjusted to 4.1, amount of NaCl was 1 mol L-1 and extraction process was performed at a temperature of 45 0C. The linear ranges were 5-250 μg L-1 for 3, 6-NDS-1-OH (1-naphthol-3,6-disulfonic acid), 2-NS (2-naphthalene sulfonate) and 1-NS and 10- 250 μg L-1 for 5-NH2-1-NS (5-Amino-1-naphthalenesulfonic acid). The detection limits were within the range of 1.20- 2.97 μg L-1. Under the optimum conditions, with the enrichment factor of the NSs were 27, 10, 14 and 7 fold for 2-NS, 1-NS, 3, 6- NDS-1-OH and 5-NH2-1-NS respectively. The RSDs was within 1–9.4%. The method is very suitable for the determination of naphthalene sulfonates (NSs) in seawater samples.
References
- E. Caro, R. M. Marcé, P. A. G, Cormack, D. C. Sherrington, F. Borrull, J. Chromatogr. A 1047, 175, (2004).
- T. O. Hanci, Z. Kartal, I. A. Alaton, J. Environ. Manage. 99, 44, (2012).
- I. A. Alaton, T. O. Hanci, Environ. Pollution 16, 413, (2009).
- C. H. Liu, W. H. Ding, J. Chromatogr. A 49, 349, (2002).
- R. A. Gimeno, J. L. Beltrán, R. M. Marcé, F. Borrull, J. Chromatogr. A 890, 289, (2000).
- V. Gianotti, F. Gosetti, S. Polati, M. C. Gennaro, Chemospher. 67, 1993, (2007).
- C. H. Liu, W. H. Ding, J. Chromatogr. A 926, 341, (2001).
- M. Nottebohm, T. Licha, J. Chromatogr. Sci. 50, 477, (2012).
- S. H. Hashemi, M. Kaykhaii, M. Khajeh, Anal. Lett. 48, 1815, (2015).
- M. Khajeh, M. Bohlooli, H. Hashemi, J. Macromol. Sci. 46, 1, (2009).
- M. Khajeh, M. Kaykhaii, M. Mirmoghaddam, H. Hashemi, Intern. J. Environ. Anal. Chem. 89, 981, (2009).
- M. Kaykhaii, M. Khajeh, S. H. Hashemi, J. Anal. Chem. 70, 1325, (2015).
- S. H. Hashemi, M. Khajeh, M. Kaykhaii, Anal. Methods 5, 2778, (2013).
- M. Khajeh, M. Kaykhaii, H. Hashemi, M. Mirmoghaddam, polym. Sci. Ser. B. 51, 344, (2009).
- M. Kaykhaii, G. W. Dicinoski, P. R. Haddad, Anal. Lett. 43, 1546, (2010).
- S. Risticevic, V. H. Niri, D. Vuckovic, J. Pawliszyn. Anal. Bioanal. Chem. 393, 781, (2009).
- W. M. Mullett, P. Martin, J. Pawliszyn, Anal. Chem. 73, 2383, (2001).
- J. P. Lambert, W. M. Mullett, E. Kwong, D. Lubda, J. Chromatogr. A. 1075, 43, (2005).
- Z. Xu, Y. Hu, Y. Hu, G. Li, J. Chromatogr. A. 1217, 3612, (2010).
- Y. Hu, J. Li, Y. Hu, G. Li, Talanta. 82, 464, (2010).
- N. Zhang, B. Hu, Anal. Chim. Acta. 723, 54, (2012).


