DEVELOPMENT OF AN ANALYTICAL METHOD FOR THE MAIN ORGANIC COMPOUNDS DERIVED FROM THERMOCHEMICAL CONVERSION OF BIOMASS
- Liquid chromatograph,
- ,
- Organic acids,
- Levoglucosan,
- Solid phase extraction
- Aqueous bio-oil,
- Liquid HTC process ...More
Copyright (c) 2016 Catherine Tessini, Romina Romero, Mauricio Escobar, Alfredo Gordon, Mauricio Flores

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Abstract
In this work, high-performance liquid chromatography (HPLC-UV/RID) is applied to the simultaneous determination of acetic acid, formic acid, acetol, glyoxal, glycolaldehyde and levoglucosan in a by-product in an aqueous liquid phase that is produced by the Hydrothermal Carbonization (HTC) process and in an aqueous bio-oil phase, which comes from a fast pyrolysis process. Both processes were run in forest biomass. For the development and optimization of the proposed method, some chromatographic columns were evaluated based on separation principles of reversed phase and ionic exclusion, although it was previously performed with a solid phase extraction (SPE) process. Concentrations of acetic and formic acids in the liquids of the HTC process ranged from 0.26 to 1.5 % and from 0.14 to 2.7 %, respectively. Concentrations of acetic and formic acids, levoglucosan and glycolaldehyde in the aqueous bio-oil phases ranged from 0.4 – 4.6 %, 0.4 – 1.4 %, 0.13 – 2.5 % and 0.5 – 3.5%, respectively.
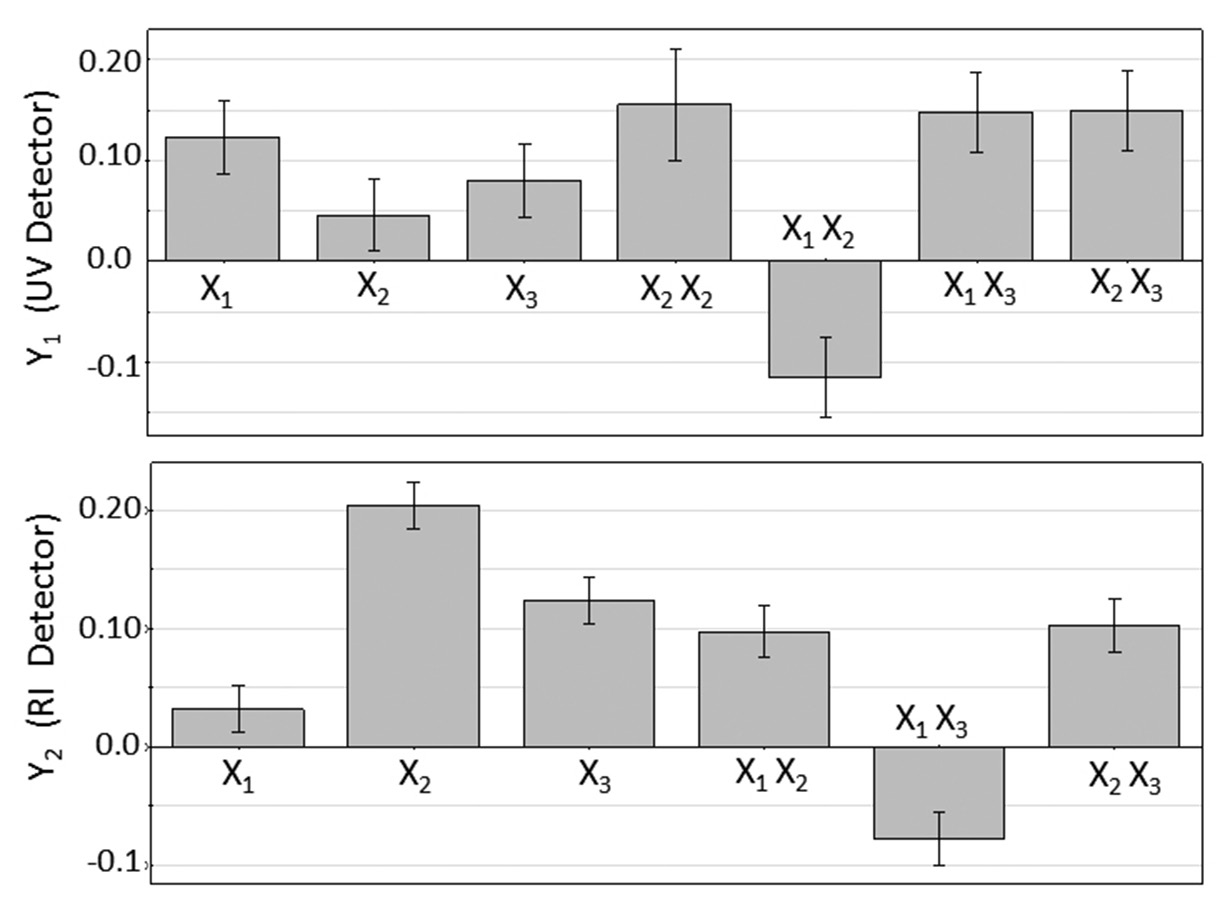
References
- -M. Guiotokua, C.R. Rambob, F.A. Hansela, W.L.E. Magalhãesa, D. Hotzab, Microwave-assisted hydrothermal carbonization of lignocellulosic materials, Mater. Lett., 63 (2009) 2707-2709.
- -H. Kambo, A. Dutta, Strength, storage, and combustion characteristics of densified lignocellulosic biomass produced via torrefaction and hydrothermal carbonization, Appl. Energ., 135 (2014) 182-191.
- -Z. Liu, F.S. Zhang, J. Wu, Characterization and application of chars produced from pinewood pyrolysis and hydrothermal treatment, Fuel, 89 (2010) 510-514.
- -J. Libra, K. Ro, C. Kammann, A. Funke, N. Berge, Y. Neubauer, M.M. Titirici, C. Fühner, O. Bens, J. Kern and K. Emmerich, Hydrothermal carbonization of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis, Biofuels 2 (2011) 89-124.
- -A. Funke, F. Ziegler, Hydrothermal carbonization of biomass: A summary and discussion of chemical mechanisms for process engineering, Biofuels, Bioprod. Bioref., 4 (2010), 160–177.
- -A. Kruse, A. Funke, M.M. Titirici, Hydrothermal conversion of biomass to fuels and energetic materials, Curr. Opin. Chem. Biol., 17 (2003) 515– 521.
- -S. Hoekman, A. Broch, C. Robbins, Hydrothermal Carbonization (HTC) of Lignocellulosic Biomass, Energ. Fuel., 25 (2011) 1802–1810.
- -Z. Qi, C. Jie, W. Tiejun, X. Ying, Review of biomass pyrolysis oil properties and upgrading research Energy Convers. Manage. 48 (2007) 87-92.
- -J.P. Diebold, National renewable energy laboratory, NREL/SR-570- 27613, 2000.
- -D. Mohan, C.U. Pittman, P.H. Steele, Pyrolysis of Wood/Biomass for Bio-oil: A Critical Review, Energ. Fuel., 203 (2006) 848−889.
- -A. Bridgwater, S. Czernik, J. Diebold, D. Meier, A. Oasmaa, C. Peacocke, J. Piskorz, D. Radlein, Fast Pyrolysis of Biomass: A Handbook, Aston University, Birmingham, 2008.
- -A.A. Boateng, C.A. Mullen, Fast pyrolysis of biomass thermally pretreated by torrefaction, J. Anal. Appl. Pyrolysis, 100 (2013) 95-102.
- -M. Asadullah, A. Mohammad, N. Suhada, N. Hanina, M. Ilmam, A. Azdarpour, Optimization of palm kernel shell torrefaction to produce energy densified bio-coal, Energ. Convers. Manage., 88 (2014) 1086– 1093.
- -S. Ren, H. Lei, L. Wanga, Q. Bu, S. Chen, J. Wua, J. Julson, R. Ruan, The effects of torrefaction on compositions of bio-oil and syngas from biomass pyrolysis by microwave heating, Bioresour. Technol., 135 (2013) 659–664.
- -M. Pelaez-Samaniegoa, V. Yadamac, M. Garcia-Pereza, E. Lowelle, A.G. McDonald, Effect of temperature during wood torrefaction on the formation of lignin liquid intermediates, J. Anal. Appl. Pyrolysis, 109 (2014) 222–233.
- -Z. Yang, M. Sarkar, A. Kumar, J. Tumuluru, R.L. Huhnke, Effects of torrefaction and densification on switchgrass pyrolysis products, Bioresour. Technol., 174 (2014) 266-273.
- -Y. Choia, P. Johnston, R Brown, B. Shanksa, K-H. Lee, Detailed characterization of red oak-derived pyrolysis oil: Integrated use of GC, HPLC, IC, GPC and Karl-Fischer, J. Anal. Appl. Pyrolysis, 110 (2014) 147–154.
- -Satyanarayan Naik, Vaibhav V. Goud, Prasant K. Rout, Ajay K. Dalai, Supercritical CO2 fractionation of bio-oil produced from wheat–hemlock biomass, Bioresource Technol., 101 (2010) 7605–7613.
- -C. Mullen, A. Boateng, Chemical Composition of Bio-oils Produced by Fast Pyrolysis of Two Energy Crops, Energ. Fuel., 22 (2008) 2104–2109.
- -A. Zotou, Z. Loukou, O. Karava, Method Development for the Determination of Seven Organic Acids in Wines by Reversed-Phase High Performance Liquid Chromatography, Chromatographia, 60 (2004) 39–44.
- -S. Deshmukh, A. Frolov, A. Marcillo, C. Birkemeyer, Selective removal of phosphate for analysis of organic acids in complex samples, J. Chromatogr. A, 1388 (2015) 1–8.
- -C. Rodrigues, L. Marta, R. Maia, M. Miranda, M. Ribeirinho, C. Máguas, Application of solid-phase extraction to brewed coffee caffeine and organic acid determination by UV/HPLC, J. Food. Compos. Anal., 20 (2007) 440–448.
- -F. Chinnici, U. Spinabelli, C. Riponi, A. Amati, Optimization of the determination of organic acids and sugars in fruit juices by ion-exclusion liquid chromatography, J. Food. Compos. Anal., 18 (2005) 121–130.
- -J. Ma, B. Zhang, Y. Wang, X. Hou, Comparison of Six Sample Preparation Methods for Analysis of Food Additives in Milk Powder, Food Anal. Method., 7 (2014) 1345-1352.
- -D. Han, M. Tian, D.W. Park, K.H. Row, Determination of organic acids in Salicornia herbacea by solid-phase extraction combined with liquid chromatography, Nat. Prod. Commun., 8 (2013) 203-206.
- -G. Xiang, L. Yang, X. Zhang, H. Yang, Z. Ren, M. Miao, A Comparison of Three Methods of Extraction for the Determination of Polyphenols and Organic Acids in Tobacco by UPLC–MS–MS, Chromatographia, 70 (2009) 1007–1010.
- -K. Hrobonova, J. Lehotay, J. Cizmarik, Determination of Organic Acids in Propolis by HPLC Using Two Columns with an On-Line SPE System, J. Liq. Chromatogr. R. T., 32 (2009) 125-135.
- -W. Xu, L. Liang, M. Zhu, Determination of Sugars in Molasses by HPLC Following Solid-Phase Extraction, Int. J. Food Prop., 18 (2015) 547-557.
- -A. de Villiers, F. Lynen, A. Crouch, P. Sandra, Development of a Solid- Phase Extraction Procedure for the Simultaneous Determination of Polyphenols, Organic Acids and Sugars in Wine, Chromatographia, 59 (2004) 403-409.
- -M. Castellari, E. Sartini, U. Spinabelli, C. Riponi, S. Galassi, Determination of Carboxylic Acids, Carbohydrates, Glycerol, Ethanol, and 5-HMF in Beer by High-Performance Liquid Chromatography and UV–Refractive Index Double Detection, J. Chromatogr. Sci., 39 (2001) 235-8.
- -J. Arsenault, Beginner’s Guide to SPE, Waters Corporation, (2012).
- -Guidelines for Single Laboratory Validation of Chemical Methods for Dietary Supplements and Botanicals, www.aoac.org, 2012, pp. 18-19.
- -J. Miller, J. Miller, Statistics and Chemometrics for Analytical Chemistry, sixth ed, Calibration methods in instrumental analysis: regression and correlation, Pearson Education Limited, Edinburgh, 2010, pp. 110–150.
- -A. Oasmaa, D. Meier, Norms and standards for fast pyrolysis liquids1. Round robin test, J. Anal. Appl. Pyrolysis 73 (2005) 323–334.
- -R. Becker, U. Dorgerloh, E. Paulke, J. Mumme, I. Nehls, Hydrothermal Carbonization of Biomass: Major Organic Components of the Aqueous Phase, Chem. Eng. Technol. 37 (2014) 511–518.
- -Tessini. C, M. Vega, N. Müller, L. Bustamante, D. von Baer, A. Berg, C. Mardones, High performance thin layer chromatography determination of cellobiosan and levoglucosan in bio-oil obtained by fast pyrolysis of sawdust, J. Chromatogr. A, 1218 (2011) 3811–3815.
- -A. Azeez, D. Meier, J. Odermatt, T. Willner, Fast Pyrolysis of African and European Lignocellulosic Biomasses Using Py-GC/MS and Fluidized Bed Reactor, Energ. Fuel., 2010, 24, 2078–2085.


