SYNTHESIS, CHARACTERIZATION, THEORETICAL AND BIOLOGICAL STUDIES OF A NEW MACROCYCLE SCHIFF BASE WHITH Co(II), Ni(II), Cu(II) AND Zn(II) COMPLEXES
- Macrocycle Schiff base,
- DFT calculations,
- thermal analysis,
- molecular modeling,
- antibacterial activity
Copyright (c) 2018 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
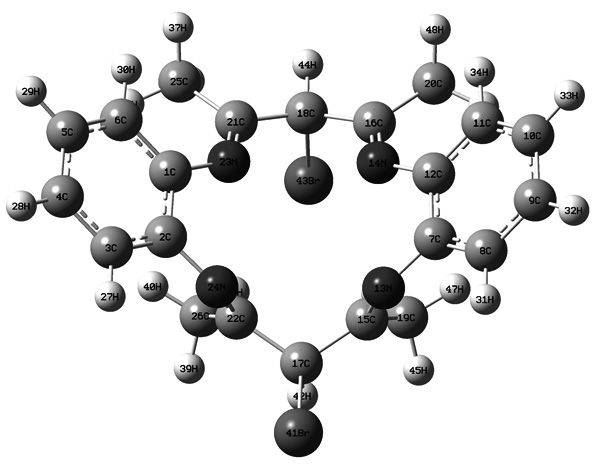
A new macrocycle Schiff base, by the reaction between benzene-1,2-diamine and 3-bromo-pentane-2,4-dione, is introduced and a series of its complexes with Co(II), Ni(II), Cu(II) and Zn(II) have been synthesized. FTIR, 1H and 13C NMR, and TGA, also biological activities of all complexes and the electronic spectral data have been investigated. The results show that there are no water molecules, also that macrocycle Schiff base is bonded to the metal ions through nitrogen atoms of four azomethine groups via an octahedral arrangement containing N4 tetra-dentate donor sites on planar and two Cl atoms occupying the other two opposed sites. The corresponding Cu(II) and Zn(II) complexes showed good antibacterial and antifungal activities. The molecular geometry, frontier molecular orbitals and Mulliken atomic charge of the compounds are investigated theoretically using DFT method.
References
- V. Mishra, S. N. Pandeya, S. Anathan, Acta Pharm. Turc. 2000, 42, 139– 148.
- A. Nishinaga, H. Tomita, K. Nishizawa, T. Matsuura, S. Ooi, K. Hirotsu, J. Chem. Soc., Dalton Trans. 1981, 1504–1514.
- B. J. Motekaitis, A. E. Martell, D. A. Nelson, Inorg. Chem. 1984, 23, 275– 283.
- M. Wang, L. F. Wang, Y. Z. Li, Q. X. Li, Z. D. Xu, D. Q. Qu, Transition Met. Chem. 2001, 26, 307–310.
- S. Park, V. K. Mathur, R. P. Planalp, Polyhedron 1998, 17, 325–330.
- X. Ran, L. Wang, Y. Lin, J. Hao, D. Cao, Appl. Organomet. Chem. 2010, 24, 741–747.
- N. Aggarwal, R. Kumar, P. Dureja, D. S. Rawat, J. Agric. Food. Chem. 2009, 57, 8520–8525.
- Y. W. Ren, H. Guo, C. Wang, J. J. Liu, H. Jiao, J. Li, F. X. Zhang, Transition Met. Chem. 2006, 31, 611–615.
- M. Salavati-Niasari, M. Rezai-Adaryni, S. Heydarzadeh, Transition Met. Chem. 2005, 30, 445–450.
- E. C. Constable, Coordination chemistry of macrocyclic compounds, University Press, Oxford, 1999.
- D. P. Singh, R. Kumar, V. Malik, P. Tyagi, Transition Met. Chem. 2007, 32, 1051–1055.
- D. P. Singh, R. Kumar, V. Malik, P. Tyagi, J. Enz. Inhib. Med. Chem. 2007, 22, 177–182.
- R. V. Singh, A. Chaudhary, J. Inorg. Biochem. 2004, 98, 1712–1721.
- A. Mobinikhaledi, M. Jabbarpour, A. Hamta, J. Chil. Chem. Soc. 2011, 56, 812-814.
- S. M. Abdallah, M. A. Zayed, G. G. Mohamed, Arab. J. Chem. 2010, 3, 103–113.
- Q.-B. Li, L.-W. Xue, W.-C. Yang, G.-Q. Zhao, J. Chil. Chem. Soc. 2013, 58, 1880-1883.
- D. P. Singh, R. Kumar, M. Kamboj, K. Jain, Acta Chim. Slov. 2009, 56, 780–785.
- S. Han, Y. Wang, J. Chil. Chem. Soc. 2014, 59, 2753-2755.
- G. G. Mohamed, M. A. Badawy, M. M. Omar, M. M. Nassar, A. B. Kamel, Spectrochim. Acta A 2010, 77, 773-781.
- W. Li, Y. Ding, X.-S. Cheng, Z. You, J. Chil. Chem. Soc. 2015, 60, 3034- 3037.
- M. Shakir, N. Shahid, N. Sami, M. Azam, A. U. Khan, Spectrochim. Acta A 2011, 82, 31–33.
- A. Reiss, A. Samide, G. Ciobanu, I. Dabuleanu, J. Chil. Chem, Sic. 2015, 60, 3074-3079.
- R. N. Prasad, S. Gupta, J. Serb. Chem. Soc. 2002, 67, 523–530.
- M. B. Ferrari, C. Pelizzi, G. Pelosi, M. C. Rodrı́guez-Argüelles, Polyhedron 2002, 21, 2593–2599.
- D. P. Singh, R. Kumar, P. Tyagi, Transition Met. Chem. 2006, 31, 970–973.
- A. Chaudhary, R. Swaroop, R. Singh, Bol. Soc. Chile Quím. 2002, 47, 203–211.
- A. D. Becke, J. Chem. Phys. 1993, 98, 5648–5652.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, J. T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyenga, J. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, Gaussian 03, Revision A.1. Pittsburgh, PA, Gaussian Inc. 2007.
- R. J. Parr, R. G. Pearson, J. Am. Chem. Soc. 1983, 105, 7512–7516.
- S. Khan, S. A. A. Nami, K. S. Siddiqi, Spectrochim. Acta A, 2007, 68, 269–274.
- B. H. M. Mruthyunjayaswamy, B. I. Omkar, Y. Jadegoud, J. Brazil. Chem. Soc. 2005, 16, 783–789.
- W. G. Geary, Coord. Chem. Rev. 1971, 7, 81–122.
- K. Nakamoto, Infrared and Raman spectroscopy of inorganic and coordination compounds, Wiley-Interscience, New York, 1978.
- M. Salavati-Niasari, F. Davar, Inorg. Chem. Commun. 2006, 9, 175–179.
- P. S. Kalsi, Spectroscopy of organic compounds, New Age International (P) Ltd., New Delhi, 1999.
- J. R. Dyer, Application of absorptions spectroscopy of organic compounds, Prentice Hall, New Delhi, 1991.
- S. Sarkar, K. Dey, Spectrochim. Acta A 2010, 77, 740–748.
- J. Huang, S. A. Li, D. F. Li, D. X. Yang, W. Y. Sun, W. X. Tang, Bioorg. Med. Chem. 2004, 12, 529–535.
- P. E. Aranha, M. P. Doe Santos, S. Romera, E. R. Dockal, Polyhedron 2007, 26, 1373–1382.
- I. Demir, M. Bayrakci, K. Mutlu, A. I. Pekacar, Acta Chim. Slov. 2008, 55, 120–124.
- R. C. Maurya, P. Patel, S. Rajput, Synth. React. Inorg. Met.-Org. Chem. 2003, 33, 817–836.
- S. Arunachalam, N. P. Priya, C. Jayabalakrishnan, V. Chinnusamy, Spectrochim. Acta A 2009, 74, 591–596.
- M. S. Refat, S. A. El-Korashy, D. N. Kumar, A. S. Ahmed, Spectrochim. Acta A 2008, 70, 898–906.
- F. A. Cotton, G. Wilkinson, C. A. Murillo, M. Bochmann, Advanced inorganic chemistry, 6th ed., Wiley-Interscience, New York, 1999.
- D. Sutton, Electronic spectra of transition metal complexes, McGraw-Hill, London, 1968.
- A. B. P. Lever, Inorganic electronic spectroscopy, 2nd ed., Elsevier, Amsterdam, 1984.
- S. Chattopadhyay, M. G. B. Drew, A. Ghosh, Polyhedron 2007, 26, 3513– 3522.
- S. Banerjee, J. Gangopadhyay, C. Z. Lu, J. T. Chen, A. Ghosh, Eur. J. Inorg. Chem. 2004, 2533–2541.
- G. L. Miessler, D. A. Tarr, Inorganic chemistry, 3rd ed., Pearson prentice Hall, London, 1994.
- B. E. Douglas, D. H. McDaniel, J. J. Alexander, Concepts and models of inorganic chemistry, 3rd ed., John Wiley and Sons Inc., New York, 1994.
- M. Salavati-Niasari, F. Davar, Polyhedron 2006, 25, 2127–2134.
- S. Srivastava, A. Kalam, Synth. React. Inorg. Met.-Org. Chem. 2004, 34, 1529–1541.
- M. M. Omar, G. G. Mohamed, A. A. Ibrahim, Spectrochim. Acta A 2009, 73, 358–369.
- F. Rafat, K. S. Siddiqi, J. Korean Chem. Soc. 2011, 55, 912–918.
- L. Mallesha, K. N. Mohana, J. Chem. Pharm. Res. 2007, 2, 75–82.
- H. Keypour, M. Shayesteh, M. Rezaeivala, F. Chalabian, L. Valencia, Spectrochim. Acta A 2013, 101, 59–66.
- O. A. El-Gammal, M. M. Bekheit, S. S. El-Brashy, Spectrochim. Acta A 2015, 137, 207–219.
- S. Mondal, S. Naskar, A. K. Dey, E. Sinn, C. Eribal, S. R. Herron, S. K. Chattopadhyay, Inorg. Chim. Acta 2013, 398, 98–105.
- M. Gaber, H. El-Ghamry, F. Atlam, S. Fathalla, Spectrochim. Acta A 2015, 137, 919–929. 3740.


