DETERMINATION OF pKa AND pKb FROM ELECTRONIC PROPERTIES DERIVED FROM CONCEPTUAL DENSITY FUNCTIONAL THEORY (cDFT)

- Acidity,
- Basicity,
- Nucleophilicity,
- Electrophilicity,
- Multilinear Correlation
Copyright (c) 2024 JCChemS

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
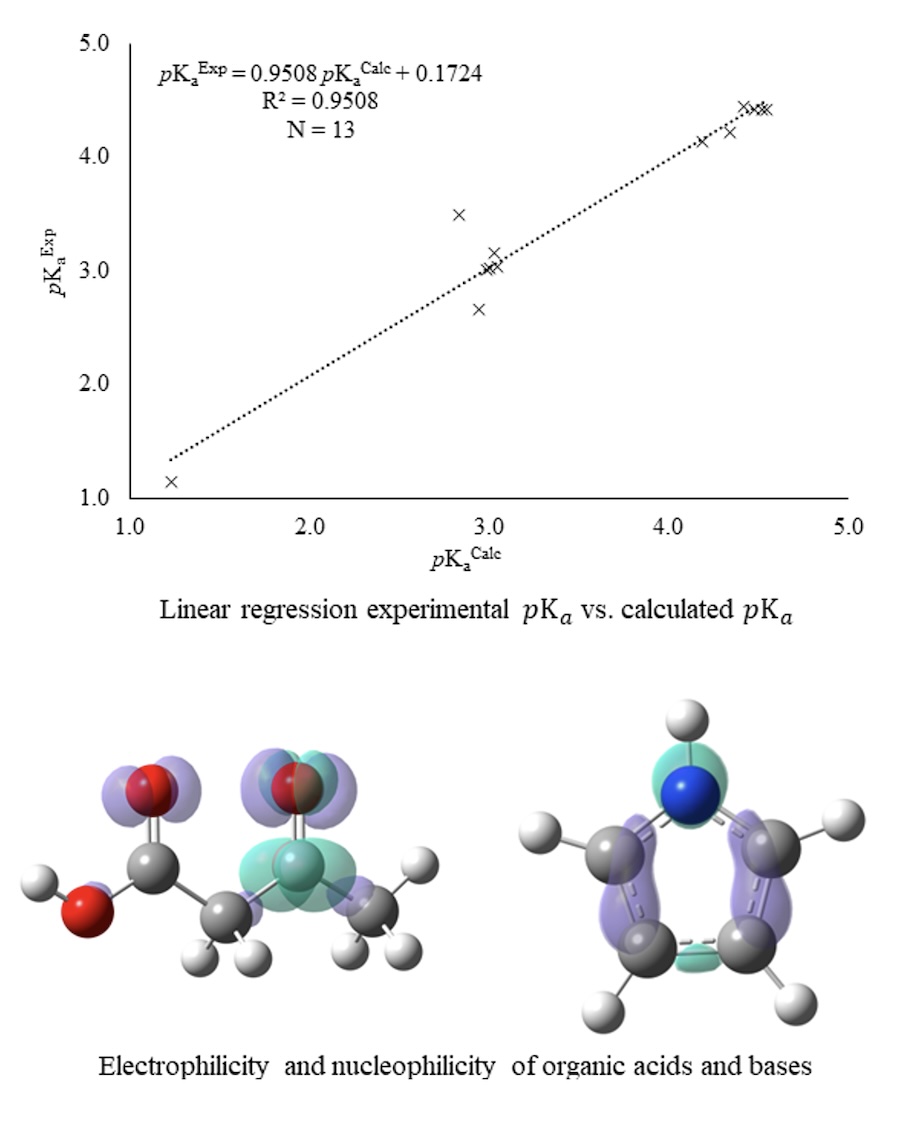
In the present work we apply a semiempirical method capable of calculating the pka and pkb values of a series of organic acids and bases through their electronic properties. This multilinear model, analogous to the one introduced by Kamlet-Taft, relates acid-base properties such as HBA and HBD to the regional electrophilicity and nucleophilicity derived from cDFT. To test the model, it was applied to a series of mono and dicarboxylic acids, as well as aliphatic/cyclic and heterocyclic amines, showing us that the model is only functional when the series of compounds present similar characteristics such as functional groups. Based on the regionalization of the electrophilicity and nucleophilicity, it can be observed that the acid-base characteristics are not exclusive to limited regions of the molecules, but that these present a bifunctional character that shifts to the relative characteristics of acid and/or base according to the global distribution of the electronic density.
References
- Löfqvist, J. Journal of Insect Physiology, 22(10), 331–1346 (1976)
- Tutusaus, O., Ni, Ch., Szymczak, N. K. Journal of the American Chemical Society, 135(9), 3403–3406 (2013)
- Lémery, L. Mém. Acad. R. Sci. (Paris), 1717 ; pp. 31–51, 122–146
- Rouelle, G. F. Mémoire sur les sels neutres. Mém. Acad. R. Sci. (Paris), 1754 ; pp. 572–588
- Bell, R.P. Acids, Bases, and the Nature of the Hydrogen Ion. In: The Proton in Chemistry. Springer, Boston, MA.
- Arrhenius, S. Philosophical Magazine Series 5, 41(251), 237–276 (1896)
- Brønsted, J. N. Einige Bemerkungen über den Begriff der Säuren und Basen, 42(8), 718–728 (1923)
- Lowry, T.M. chemistry and industry, 42(3), 43–47 (1923)
- Lewis, G.N. Valence and the Structure of Atoms and Molecules. American Chemical Monograph Series, the Chemical Catalog Co., Inc., New York, 1923 ; pp. 141-142
- Koopmans, T. Über die Zuordnung von Wellenfunktionen und Eigenwerten zu den Einzelnen Elektronen Eines Atoms. 1(1-6), 104–113 (1934).
- Fukui, K., Yonezawa, T., Shingu, H. The Journal of Chemical Physics, 20(4), 722–725 (1952)
- Trummal, A., Lipping, L., Kaljurand, I., Koppel, I. A., Leito, I. The Journal of Physical Chemistry A, 120(20), 3663-3669, (2016)
- Paresh Chandra, R., Das Kumar, P. The Journal of Physical Chemistry, 99(51), 17891–17895, (1995)
- Kütt, A., Selberg, S., Kaljurand, I., Tshepelevitsh, S., Heering, A., Darnell, A., Kaupmees, K., Piirsalu, M., Leito, I. Tetrahedron Letters, 59(42), 3738-3748, (2018)
- Bernhardsen, Ida M., Krokvik, I. R.T., Perinu, C., Pinto, D., Jens, K. J., Knuutila, H. K. International Journal of Greenhouse Gas Control, 68, 68–76, (2018)
- Manallack, David T. Perspectives in Medicinal Chemistry, 1, 25-38, (2007)
- Petukh, M., Stefl, S. Alexov, E. Current Pharmaceutical Design, 19(23), 4182–4190, (2013)
- Ullrich, K, J. Specificity of transporters for ‘organic anions’ and ‘organic cations’ in the kidney, 1197(1), 45–62, (1994)
- Reijenga, J., van Hoof, A., van Loon, A., Teunissen, B. Analytical Chemistry Insights, 8, 53–71, (2013)
- Romero, R., Salgado, P. R., Soto, C., Contreras, D., Melin, V. Frontiers in Chemistry, 6, 208, (2018)
- Gift, A. D., Stewart, S. M., Kwete Bokashanga, P. Journal of Chemical Education, 89(11), 1458–1460, (2012)
- Jia, Z., Du, D., Zhou, Z., Zhang, A., & Hou, R. Chemical Physics Letters, 439(4-6), 374–380, (2007)
- Thapa, B. & Schlegel, H. B. The Journal of Physical Chemistry A, 120(28), 5726–5735, (2016)
- Dutra, F. R., Silva, C. de S., & Custodio, R. The Journal of Physical Chemistry A, 125(1), 65–73, (2020)
- Sandoval-Lira, J., Mondragón-Solórzano, G., Lugo-Fuentes, L. I., & Barroso-Flores, J. Journal of Chemical Information and Modeling, 60, 1445-1452, (2020)
- Dahmani, R., Manachou, M., Belaidi, S., Chtita, S., & Boughdiri, S. New Journal of Chemistry, 45(3), 1253–1261, (2021)
- Panayiotou, C., Mastrogeorgopoulos, S., & Hatzimanikatis, V. The Journal of Chemical Thermodynamics, 110, 3–15, (2017)
- Kamlet, M. J., Doherty, R. M., Abraham, M. H., Marcus, Y., & Taft, R. W. The Journal of Physical Chemistry, 92(18), 5244–5255, (1988)
- Kamlet, M. J., & Taft, R. W. Journal of the American Chemical Society, 98(2), 377–383, (1976)
- Taft, R. W., & Kamlet, M. J. Journal of the American Chemical Society, 98(10), 2886–2894, (1976)
- Parr, R. G., Weitao, Y. Density – Functional Theory of Atoms and Molecules. Oxford University Press, USA Chap. 4 – 5, 1994 ; pp. 70 – 104
- Pearson, R. G. Journal of Chemical Sciences, 117(5), 369–377, (2005)
- Parr, R. G., Szentpály, L. v., & Liu, S. Journal of the American Chemical Society, 121(9), 1922–1924, (1999)
- Parr, R. G., & Yang, W. Journal of the American Chemical Society, 106(14), 4049–4050, (1984)
- Hohenberg, P., & Kohn, W. Physical Review, 136(3B), B864–B871, (1964)
- Janak, J. F. Physical Review B, 18(12), 7165–7168, (1978)
- Contreras, R., Andres, J., Safont, V. S., Campodonico, P., Santos, J. G. The Journal of Physical Chemistry A, 107(29), 5588–5593, (2003)
- Politzer, P., Murray, J. S., Bulat, F. A. A review, 16(11), 1731–1742, (2010)
- Weiß, N., Schmidt, C. H., Thielemann, G., Heid, E., Schröder, C., & Spange, S. Physical Chemistry Chemical Physics, 23(2), 1616–1626, (2021)
- Vela, A., & Gazquez, J. L. Journal of the American Chemical Society, 112(4), 1490–1492, (1990)
- Simón-Manso, Y., & Fuentealba, P. The Journal of Physical Chemistry A, 102(11), 2029–2032, (1998)
- Contreras, R. R., Fuentealba, P., Galván, M., & Pérez, P. Chemical Physics Letters, 304(5-6), 405–413, (1999)
- Cerda-Monje, A., Ormazábal-Toledo, R., Cárdenas, C., Fuentealba, P., & Contreras, R. The Journal of Physical Chemistry B, 118(13), 3696–3701, (2014)
- Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Scalmani, G., Barone, V., Mennucci, B., Petersson, G. A., et al. Gaussian 09, Revision E.01; Wallingford, CT, 2013
- Dawson, R.M.C., Elliot, D.C., Elliot, W.H. and Jones, K. M. Data for biochemical research. Clarendon Press, Oxford, (1986)
- Brown, H. C., McDaniel, D. H., & Hafiiger, O. Determination of Organic Structures by Physical Methods," ed. by Braude E. A. and Nachod, F. C., Academic Press, New York, 1959
- Lide, D. R. Handbook of Chemistry and Physics. Edition 84°, CRC press, 2004
- Chakkamalayath, J., Kuttay, C. R. S., & Ghosh, S. K. The Journal of Physical Chemistry A, 124, 3770-3777, (2020)
- Barret, R. Therapeutical Chemistry, 21–51 (2018)
- Siegel, A. F. & Wagner, M. R. Chapter 12 - Multiple Regression: Predicting One Variable from Several Others, Editor(s): Andrew F. Siegel, Michael R. Wagner, Practical Business Statistics (Eighth Edition), Academic Press, 2022 ; pp. 371-431.

