- Absolute configuration,
- Diterpenoid,
- X-ray diffraction,
- Crystal and Supramolecular structure
Copyright (c) 2017 Iván Brito, Jorge Borquez, Alejandro Cárdenas, Maria Avendaño, Mario Simirgiotis

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
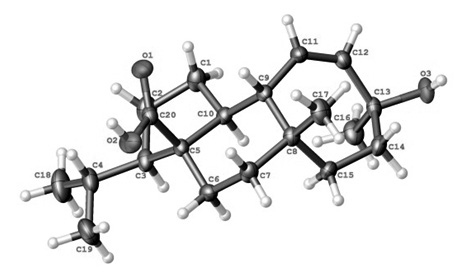
The molecular structure of the title compound [iupac name: 13-epi-8–hydroxy-3-isopropyl-5a,8-dimethyl-2,3,4,5,5a,6,7,8,10a,10b-decahydrocyclohepta[e] indene-3a(1H) acid] is similar to the structure of the known compound mulinolic acid.The structure of this mulinolic acid epimer consists of a mulinane skeleton and the isopropyl, methyl group, carboxylic acid and the hydroxyl group at C3, C8, C5, C13, respectively, which are β-oriented, whereas the methyl group at C13 are α-oriented. The cyclopentane (A), ciclohexane (B) and cicloheptene (C) rings are trans (A/B) and (B/C) cis fused, and are in an envelope, chair, and twist chair conformation respectively. In the crystal the molecules are linked by two strong intermolecular OH⋅⋅⋅O hydrogen bond forming bidimensional supramolecular structures with graph-set notation (12) and (46).The absolute configuration of the title compound which has been determined from the refinement of the Flack parameter16. On this basis the absolute configuration was assigned as C3R, C5S, C8S, C9S, C10S and C13S.
References
- L. A. Loyola, J. Bórquez, G. Morales, A. San-Martín. Phytochemistry. 44, 649, (1997).
- L. A. Loyola, J. Bórquez, G. Morales, A. San-Martín. Phytochemistry. 45, 1465, (1997).
- L. A. Loyola, G. Morales, B. Rodríguez, J. Jiménez-Barbero, S. Pedreros, M. C. de la Torre, A. Perales, M. R. Torres. Tetrahedron. 46, 5413, (1990).
- L. A. Loyola, G. Morales, B. Rodríguez, J. Jiménez-Barbero, S. Pedreros, M. C de la Torre, A. Perales. Journal of Natural Product. 54, 1404, (1991).
- L. A. Loyola, J. Bórquez, G. Morales, A. San-Martín. Phytochemistry. 43, 165, (1996).
- L. A. Loyola, J. Bórquez, G. Morales, A. San-Martín. Phytochemistry. 53, 961, (2000).
- I.Brito, M.J.Simirgiotis, A.Brito, M.R.Werner, J. Bórquez, P. Winterhalter, A. Cárdenas. Journal of the Chilean Chemical Society 60, (1), 2864, (2015).
- I. Brito, J. Bórquez, M. Simirgiotis, M. Neves-Vieira, G. Jerz, P. Winterhalter, M. Bolte, A. Cárdenas. Zeitschrift für Kristallographie-New Crystal Structures 229, 399, (2014).
- J. Bórquez, A.Ardiles, L.A. Loyola, L.M. Peña-Rodriguez, G.M. Molina- Salinas, J. Vallejos, I.G.Collado, M.J. Simirgiotis. Molecules, 19, 3898 (2014).
- J. Bórquez, N.L. Bartolucci, C. Echiburú-Chau, P. Winterhalter, J. Vallejos, G. Jerz, M.J. Simirgiotis. Journal of the Science of Food and Agriculture 96, (8), 2832 , (2016).
- J. Bórquez, N. L. Bartolucci, C. Echiburú-Chau, P. Winterhalter, J. Vallejos, G. Jerz, M. J. Simirgiotis. Journal of the Science of Food and Agriculture. 96, (8), 2832, (2016).
- L. A. Loyola, J. Bórquez, G. Morales, A.San Martín. Phytochemistry. 4, (1), 165, (1996).
- G. M. Sheldrick, Acta Cryst. 112 , A64, (2008).
- O.V. Dolomanov, L.J. Bourhis, R.J. Gildea, J.A.K. Howard, H. Puschmann, J. Appl. Cryst., 42, 339, (2009).
- D.Cremer, J.A. Pople. J.Am.Chem.Soc. 97, 1354, (1975).
- H.D. Flack. Acta Cryst. A39, 876, (1983).
- J. Bernstein, R.E. Davis, L. Shimoni, N.-L. Chang. Angew. Chem. Int. Ed. Engl. 34, 1555, (1995).


