VALIDATED STABILITY INDICATING RP-HPLC METHOD FOR SIMULTANEOUS DETERMINATION OF NORTRIPTYLINE AND PREGABALIN IN BULK AND COMBINED DOSAGE FORMULATIONS
- Nortriptyline,
- Pregabalin,
- HPLC,
- Stability indicating,
- Validation
Copyright (c) 2017 Haritha Potluri, Sreenivasa Rao Battula, Sunandamma Yeturu

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
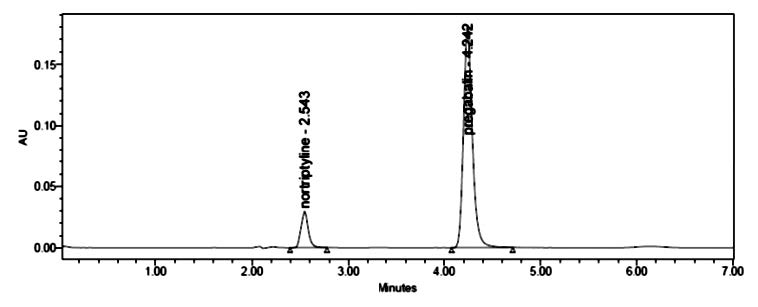
A simple and precise stability indicating RP-HPLC method was developed for simultaneous analysis of drug nortriptyline and pregabalin using BDS (250mm x 4.6 mm, 5m) C18 column at 210 nm of UV detection. Perchloric acid (0.1%) and acetonitrile in the ratio of 55:45 was used as the mobile phase with a flow rate of 1.0 ml/min and linearity response was established over the concentration range of 5-30 μg/ml for nortriptyline and 37.5-225 μg/ml for pregabalin. The active pharmaceutical ingredients recovered for nortriptyline and pregabalin are in the range of 100.60-101.65% and 100.59-101.74% respectively. The method was validated and was found to be stability indicating and can be successfully utilized for the quantitative analysis of pharmaceutical tablet dosage formulations containing nortriptyline and pregabalin.
References
- Gillman P K .Tricyclic antidepressant pharmacology and therapeutic drug interactions updated .Br. J. Pharmacol. 151, 737, (2007).
- The British Pharmacopoeia, The Stationery Office, vol. 2, London, 2010; pp. 1295–1297.
- Taylor CP, Angelotti T, Fauman E. Pharmacology and mechanism of action of pregabalin: The calcium channel alpha2-delta (alpha2-delta) subunit as a target for antiepileptic drug discovery. Epilepsy Res.73, 137, (2007).
- Indian Pharmacopoeia, vol. 3, sixth edition, Ghaziabad, 2010; pp. 1960- 1961.
- Bakshi M, Singh S. Development of validated stability indicating assay methods- critical review. J Pharm Biomed Anal. 28, 1011, (2002).
- Geneva: International Conference on Harmonization; ICH. QIA (R2) Harmonised Tripartite Guideline. Stability Testing of New Drug Substances and Products, 2003; pp. 1-18.
- Rahman H, Khatoon A, Rahman N. A validated kinetic method for determination of nortriptyline hydrochloride in pharmaceutical formulations. Int J Pharm Chem. 4, 99, (2014).
- Ashour S, Kattan N. Simultaneous determination of nortriptyline hydrochloride and fluphenazine hydrochloride in microgram quantities from low dosage forms by liquid chromatography–UV detection. J Pharm Anal. 2, 437, (2012).
- Kasawar G B, Farooqui M N .Development and validation of HPLC method for the determination of Pregabalin in capsules.Indian J Pharm Sci. 72 , 517, (2010).
- Kannapan N, Nayak S P, Venkatachalam T, Prabhakaran V. Analytical RP-HPLC method for development and validation of pregabalin and methylcobalamine in combined capsule formulation. Appl Chem Res. 13, 85, (2010).
- Mohan A J, Raj Kumar B, Bhavya T, Ashok Kumar A. RP-HPLC Method Development and validation for the simultaneous quantitative estimation of pregabalin, mecobalamin and alpha lipoic Acid in capsules. Int J Pharm Pharmsci. 6, 270, (2014).
- Gelani H D, Chauhan P P, Shah S K. Practical Implication of Chromatographic Method for Estimation of Aceclofenac and Pregabalin in Bulk and Pharmaceutical Dosage Forms. Chromatography Research International. 5, (2014).
- Pola L M, Sankar D G. Validated stability indicating RP-LC method for simultaneous quantitative estimation of aceclofenac and pregabalin in bulk drug and combined tablet dosage form. Int J Pharm. 5, 78, (2014).
- Geneva: International Conference on Harmonization; ICH. Q2 (R1) Harmonised Tripartite Guideline. Validation of Analytical Procedures: Text and Methodology, 2005; pp. 1-13.


