- Tannin, adsorption, desorption, water pollution, metal ions.
Copyright (c) 2022 SChQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
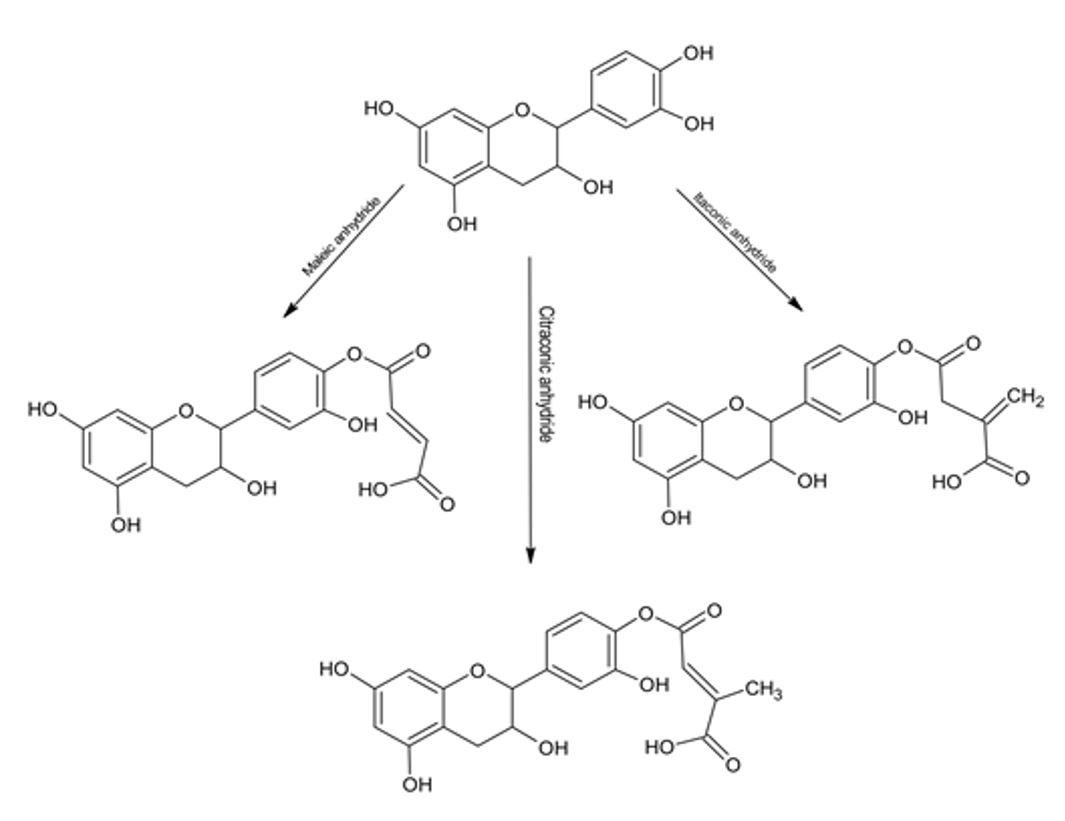
A large amount of copper ions are released into the environment, which significantly affects water resources. The reduction of copper contaminants and the potential reutilization of these ions are important issues for environmental and human health. Resin based on Pinus radiata tannin and its derivatives obtained via chemical modifications with three cyclic anhydrides (maleic, citraconic, and itaconic) adsorbed Cu(II) from aqueous solutions. The effect of the pH on the adsorption, desorption, and maximum adsorption capacity for Cu(II) solutions was studied.The adsorption depends largely on the pH of the solution, and the highest adsorption was observed at pH 3. Modified tannins showed lower maximum adsorption values than the original tannin. However, tannins modified with maleic and citraconic anhydrides presented larger values than the unmodified tannin under high ion concentrations. The resin showed a Cu(II) adsorption capacity of approximately 55 mmol g-1 and a desorption capacity higher55%, hence, it represents a good alternative for future use in the treatment of contaminated water.
References
- M.C.S. Minello, A. L. Paçó, R. S. D. Castro, L. Caetano, P. M. Padilha, G. Ferreira, M. A. U. Martines& G. R. Castro, “Evaluation of heavy metal availability in contaminated sediments from the Ilha Solteira hydroelectric dam on the Paraná River at IlhaSolteira, SP, BRAZIL,” Fresenius Environmental Bulletin, 19, 2210-2214, 2010.
- Q. Wang & Z. Yang, “Industrial water pollution, water environment treatment, and health risks in China,” Environmental Pollution, 218, 358-365, 2016.
- A. E. Evans, J. Mateo-Sagasta, M. Qadir, E. Boelee& A. Ippolito, “Agricultural water pollution: key knowledge gaps and research needs,” Current Opinion in Environmental Sustainability, 36, 20-27, 2019.
- S. Hokkanen, E. Repo, T. Suopajarvi, H. Liimatainen, J. Niinimaa& M. Sillanpaa, “Adsorption of Ni(II), Cu(II) and Cd(II) from aqueous solutions by amino modified nanostructured microfibrillated cellulose,” Cellulose, 21, 1471-1487, 2014.
- G. Z. Kyzas, G. Bomis, R. I. Kosheleva, E. K. Efthimiadou, E. P. Favvas, M. Kostoglou, & A. C. Mitropoulos, “Nanobubbles effect on heavy metal ions adsorption by activated carbon,” Chemical Engineering Journal, 356, 91-97, 2019.
- A. Piccolo, R. Spaccini, A. De Martino, F. Scognamiglio, & V. di Meo, “Soil washing with solutions of humic substances from manure compost removes heavy metal contaminants as a function of humic molecular composition,” Chemosphere, 225, 150-156, 2019.
- Y. L. Xu, S. Y. Song, J. D. Chen, R. A. Chi & J. X. Yu, “Simultaneous recovery of Cu2+ and Pb2+ from metallurgical wastewater by two tandem columns fixed respectively with tetraethylenepentamine and phosphoric acid modified bagasse,” Journal of the Taiwan Institute of Chemical Engineers, 99, 132-141, 2019.
- J. Yuan, Y. Yang, X. H. Zhou, Y. C. Ge & Q. R. Zeng, “A new method for simultaneous removal of heavy metals and harmful organics from rape seed meal from metal-contaminated farmland,” Separation and Purification Technology, 210, 1001-1007, 2019.
- Q. Zhou, B. Liao, L. Lin, Z. Song, Z. H. Khan & M. Lei, “Characteristic of adsorption cadmium of red soil amended with a ferromanganese oxide-biochar composite,” Environmental Science and Pollution Research, 26(5), 5155-5163, 2019.
- G. Palma, J. Freer & J. Baeza, “Removal of metal ions by modified Pinusradiata bark and tannins from water solutions,” Water Research, 37, 3974-3980, 2003.
- S. Hasan, T. K. Ghosh, D. S. Viswanath, & V. M. Boddu, “Dispersion of chitosan on perlite for enhancement of copper (II) adsorption capacity,” Journal of Hazardous Materials, 152(2), 826-837, 2008.
- Y. Sun, K. J. Shah, W. Sun & H. Zheng, “Performance evaluation of chitosan-based flocculants with good pH resistance and high heavy metals removal capacity,” Separation and Purification Technology, 215, 208-216, 2019.
- H. Sun, N. Xia, Z. Liu, F. Kong & S. Wang, “Removal of copper and cadmium ions from alkaline solutions using chitosan-tannin functional paper materials as adsorbent,” Chemosphere, 236, 124370, 2019.
- X. Zhao, J.C. Joo, J.K. Lee, & J.Y. Kim, “Mathematical estimation of heavy metal accumulations in Helianthus annuus L. with a sigmoid heavy metal uptake model,” Chemosphere, 220, 965-973, 2019.
- B.L. Rivas, H.A. Maturana, R.E. Catalán, I.M. Perich, “Branched and linear polyethyleneimine supports for resins with retention properties for copper and uranium. VII,” Journal of Applied Polymer Science, 38(5), 801-807, 1989.
- B.L. Rivas, H.A. Maturana, E. Pereira, “Metal ion binding properties of synthetic vinyl resins,” Die AngewandteMakromolekulareChemie: Applied Macromolecular Chemistry and Physics, 220(1), 61-74, 1994.
- B.L. Rivas, S.A. Pooley, E.D. Pereira, R. Cid, M. Luna, M. A. Jara, K. E. Geckeler, “Water‐soluble amine and imine polymers with the ability to bind metal ions in conjunction with membrane filtration,” Journal of Applied Polymer Science, 96(1), 222-231, 2005.
- J.A. Sánchez, B.L. Rivas, S.A. Pooley, L. Basaez, E.Pereira, I. Pignot-Paintrand, J. C. Moutet, “Electrocatalytic oxidation of As (III) to As (V) using noble metal–polymer nanocomposites,” ElectrochimicaActa, 55(17), 4876-4882, 2010.
- W.W Ngah, L. C. Teong, M. A. K. M. Hanafiah, “Adsorption of dyes and heavy metal ions by chitosan composites: A review,” Carbohydrate Polymers, 83(4), 1446-1456, 2011.
- B.L. Rivas, E.D. Pereira, M. Palencia, J. Sánchez, “Water-soluble functional polymers in conjunction with membranes to remove pollutant ions from aqueous solutions” Progress in Polymer Science, 36(2), 294-322, 2011.
- J. Sánchez, L. Riffo, P. Salazar, B. L. Rivas, "Removal of molybdate and vanadate ions by a copolymer adsorbent in a ultrafiltration system,” Journal of Applied Polymer Science, 136(45), 48184, 2019.
- D.V. Morales, O. Kusku, B.L. Rivas, M. Arda, N. Kabay, M. Bryjak, “Removal of Cr(VI) by stabilized solvent impregnated resin (sir) prepared by using a hydrophilic polymer adsorbent and aliquat 336,” Journal of the Chilean Chemical Society, 64(2), 4432-4436, 2019.
- D. Sud, G. Mahajan, M.P. Kaur, “Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions–A review,” Bioresource Technology, 99(14), 6017-6027, 2008.
- D.E. García, W. Glasser, A. Pizzi, C. Lacoste, M. P. Laborie, “Polyphenolic resins prepared with maritime pine bark tannin and bulky-aldehydes,” Industrial Crops and Products, 62, 84-93, 2014.
- A.P. Lim, A.Z. Aris, “A review on economically adsorbents on heavy metals removal in water and wastewater,” Reviews in Environmental Science and Bio/Technology, 13(2), 163-181, 2014.
- W. Wafwoyo, C. W. Seo, W. E. Marshall, “Utilization of peanut shells as adsorbents for selected metals,” Journal of Chemical Technology & Biotechnology: International Research in Process, Environmental & Clean Technology, 74(11), 1117-1121, 1999.
- S. Chamarthy, C.W. Seo, W.E. Marshall, “Adsorption of selected toxic metals by modified peanut shells,” Journal of Chemical Technology & Biotechnology: International Research in Process, Environmental & Clean Technology, 76(6), 593-597, 2001.
- P. Kumar, S.S. Dara, “Binding heavy metal ions with polymerized onion skin” Journal of Polymer Science: Polymer Chemistry Edition, 19(2), 397-402, 1981.
- J.M. Rodríguez-Díaz, J. O.P. García, L. R.B. Sánchez, M.G. C. da Silva, V.L. da Silva, L. E. Arteaga-Pérez, “Comprehensive characterization of sugarcane bagasse ash for its use as an adsorbent,” Bioenergy Research, 8(4), 1885-1895, 2015.
- A.M. Pintor, C.I. Ferreira, J.C. Pereira, P. Correia, S.P. Silva, V. J. Vilar& R. A. Boaventura, “Use of cork powder and granules for the adsorption of pollutants: a review,” Water Research, 46(10), 3152-3166, 2012.
- S. K. Srivastava, A. K. Singh, A. Sharma, “Studies on the uptake of lead and zinc by lignin obtained from black liquor–a paper industry waste material,” Environmental Technology, 15(4), 353-361, 1994.
- F.H. Luzardo, F.G. Velasco, C.P. Alves, I. K. D. S. Correia, L. L. Cazorla, “Chemical characterization of agroforestry solid residues aiming its utilization as adsorbents for metals in water,” RevistaBrasileira de EngenhariaAgrícola e Ambiental, 19(1), 77-83, 2015.
- K. Seki, N. Saito, M. Aoyama, “Removal of heavy metal ions from solutions by coniferous barks,” Wood Science and Technology, 31(6), 441-447, 1997.
- V. Gloaguen, H. Morvan, “Removal of heavy metal ions from aqueous solution by modified barks,” Journal of Environmental Science & Health Part A, 32(4), 901-912, 1997.
- I. Gaballah, G. Kilbertus, “Recovery of heavy metal ions through decontamination of synthetic solutions and industrial effluents using modified barks,” Journal of Geochemical Exploration, 62(1-3), 241-286, 1998.
- A. Şen, H. Pereira, M.A. Olivella, I.Villaescusa, “Heavy metals removal in aqueous environments using bark as a biosorbent,” International Journal of Environmental Science and Technology, 12(1), 391-404, 2015.
- C.W. Kim, D.S. Kim, S.Y. Kang, M. Marquez, Y. L. Joo, “Structural studies of electrospun cellulose nanofibers,” Polymer, 47(14), 5097-5107, 2006.
- P. Kumararaja, K. M. Manjaiah, S. C. Datta, T. A. Shabeer, B. Sarkar, “Chitosan-g-poly (acrylic acid)-bentonite composite: a potential immobilizing agent of heavy metals in soil,” Cellulose, 25(7), 3985-3999, 2018.
- M. Stephen, N. Catherine, M. Brenda, K. Andrew, P. Leslie, G. Corrine, “Oxolane-2, 5-dione modified electrospun cellulose nanofibers for heavy metals adsorption,” Journal of Hazardous Materials, 192(2), 922-927, 2011.
- F.H. Luzardo, F.G. Velasco, I.K. Correia, P. M. Silva, L.C. Salay, “Removal of lead ions from water using a resin of mimosa tannin and carbon nanotubes,” Environmental Technology & Innovation, 7, 219-228, 2017.
- X. Sun, X. Huang, X.P. Liao & B. Shi, “Adsorptive removal of Cu (II) from aqueous solutions using collagen-tannin resin,” Journal of Hazardous Materials, 186(2-3), 1058-1063, 2011.
- J. Freer, J. Baeza, H. Maturana, G. Palma, N. Durán, “Removal and recovery of uranium by modified Pinusradiata D. Don bark,” Journal of Chemical Technology & Biotechnology, 46(1), 41-48, 1989.
- D.E. García, N. Delgado, F.L. Aranda, M.A. Toledo, G. Cabrera-Barjas, E.M. Sintjago, S. Paczkowski, “Synthesis of maleilatedpolyflavonoids and lignin as functional bio-based building-blocks,” Industrial Crops and Products, 123, 154-163, 2018.
- D.E. García, W. G. Glasser, A. Pizzi, S. Paczkowski, M.P. Laborie, “Hydroxypropyl tannin from Pinuspinaster bark as polyol source in urethane chemistry,” European Polymer Journal, 67, 152-165, 2015.
- D.E. Garcia, W.G. Glasser, A. Pizzi, S.P. Paczkowski, M.P. Laborie, “Modification of condensed tannins: from polyphenol chemistry to materials engineering,” New Journal of Chemistry, 40(1), 36-49, 2015.
- P. Das, J. P. Claverie, “Synthesis of single‐core and multiple‐core core‐shell nanoparticles by RAFT emulsion polymerization: Lead sulfide‐copolymer nanocomposites,” Journal of Polymer Science Part A: Polymer Chemistry, 50(14), 2802-2808, 2012.
- D.E. García, P.A. Medina, V.I. Zúñiga, “Toxicological features of maleilatedpolyflavonoids from Pinusradiata (D. Don.) as potential functional additives for biomaterials design,” Food and Chemical Toxicology, 109, 1069-1078, 2017.
- D. Kołodyńska, J.A. Krukowska, P. Thomas, “Comparison of sorption and desorption studies of heavy metal ions from biochar and commercial active carbon,” Chemical Engineering Journal, 307, 353-363, 2017.
- K. G. Nierop, C. M. Preston & J. M. Verstraten, “Linking the B ring hydroxylation pattern of condensed tannins to C, N and P mineralization. A case study using four tannins,” Soil Biology and Biochemistry, 38(9), 2794-2802, 2006.
- R.T. Morrison & R. N. Boyd. “QuímicaOrgánica” 1998.
- C. Christou, A. Agapiou, R. Kokkinofta, “Use of FTIR spectroscopy and chemometrics for the classification of carobs origin,” Journal of Advanced Research, 10, 1-8, 2018.
- R. Fessenden, J. Fessenden, “QuímicaOrgánica,” 1983.
- D.E. García, W. Glasser, A. PizziPizzi, S. Paczkowski& M. Pierre Laborie, “Modification of Condensed Tannins: From Polyphenol Chemistry to Material Engineering.” New Journal of Chemistry, 40 (1), 36-49, 2015.
- D.E. García, J.C. Carrasco, J.P. Salazar, M.A. Pérez, R.A. Cancino, S.A. Riquelme, “Bark polyflavonoids from Pinusradiata as functional building – blocks for polylactic acid (PLA) – based green composites.” eXPRESS Polymer Letters 10(10), 835 – 848, 2016.
- D.E. García, J. Gavino, D. Escobar-Avello, R.A. Cancino, “Maleinatedpolyflavonoids and lignin as functional additives for three kinds of thermoplastics”. Iranian Polymer Journal. 26(4), 295-304, 2017.