
- : Antibiotics, Polymers, Oxytetracycline, Environment, Removal.
Copyright (c) 2020 SChQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
Oxytetracycline (OTC) is an antibiotic belonging to the tetracycline family, widely used in veterinary medicine due to its pharmacological characteristics and adequate cost/effectiveness ratio.
One of the problems presented by the therapeutic use of antibiotics such as OTC is that they do not metabolize and are expelled through animal excretions reaching the effluent of wastewater, rivers, and lagoons, causing accumulation and generating resistance to certain pathogens, mainly zoonotic bacteria.
The above has presented the need and the challenge of removing this type of contaminants present in aquatic systems, using more efficient economic and eco-friendly methods than the conventional ones already known. A promising alternative is removal by using polymeric materials.
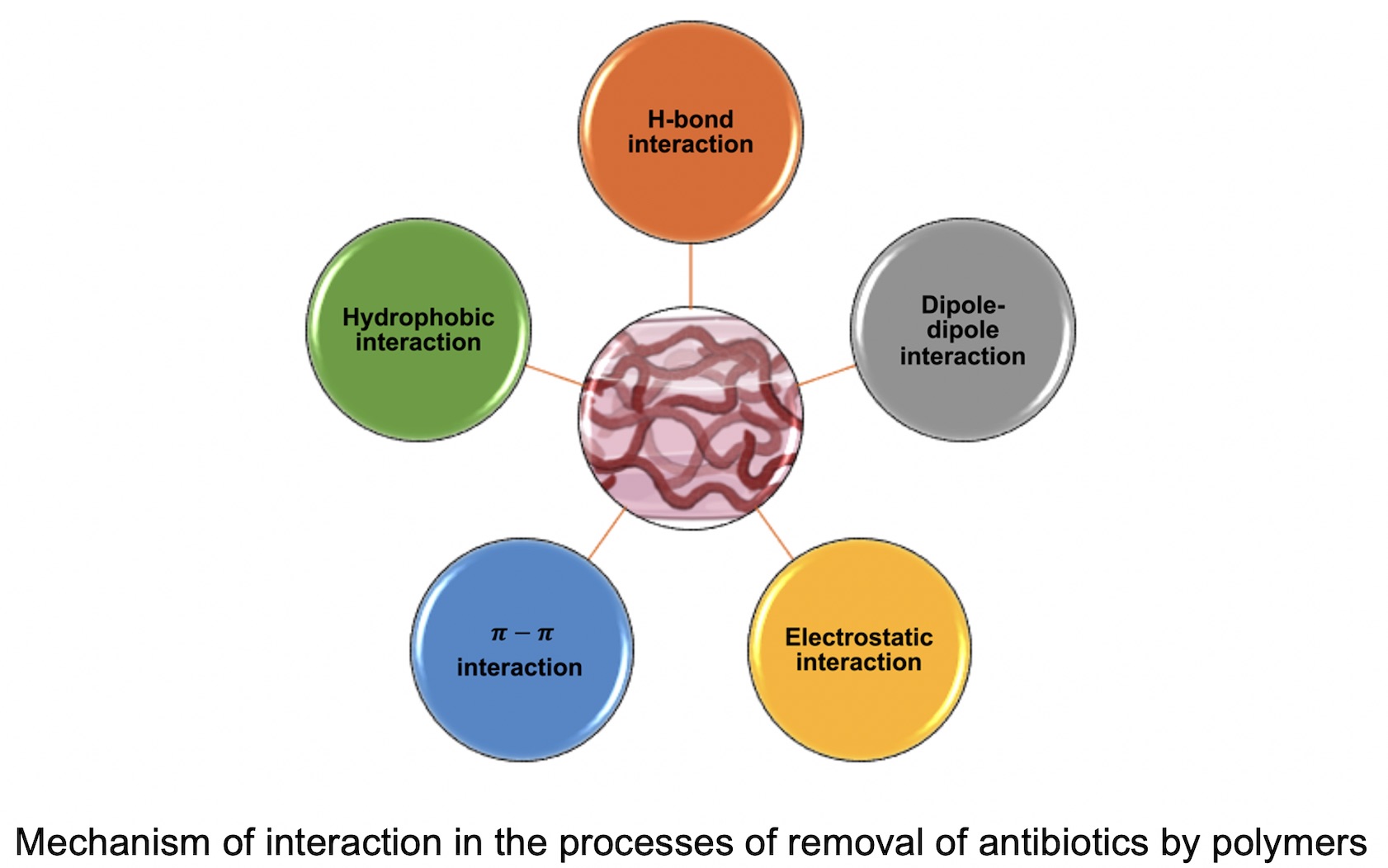
Therefore, the aim of this revision is give a general revision of the application of functional polymer materials to remove these emerging organic contaminants OTC.
References
- Cruz-Coke Madrid, R. Historia de la Medicina Chilena; Andres Bello: Santiago, Chile 1995.
- Brugueras, M. C.; García, M. M. Rev. Cuba. Med. Gen. Integr.1998, 14, 347-361.
- Morris, A.; Kellner, J. D.; Low, D. E. Curr. Opin. Microbiol. 1998, 1, 524-529.
- Doren, D.; Gabella, J. P. Terram. F. 2001, 1, 39.
- Gesche, E.; Madrid, E.; Aguila, C. Arch. Med. Vet.2001, 33, 21-29.
- Tejada, C.; Quiñonez, E.; Peña, M. Rev. Fac. Ciencias Básicas 2014, 10, 80-101.
- Gogoi, A.; Mazumder, P.; Tyagi, V. K.; Tushara Chaminda, G. G.; An, A. K.; Kumar, M. Groundw. Sustain. Dev.2018, 6, 169-180.
- Zhang, Y.; Boyd, S. A.; Teppen, B. J.; Tiedje, J. M.; Li, H. Water Res. 2014, 65, 98-106.
- Verma, A. K.; Dash, R. R.; Bhunia, P. J. Environ. Manage. 2012, 93, 154-168.
- Aljerf, L. J. Urban Environ. Eng. 2018, 12, 50-58.
- Huang, Y.; Feng, X. J. Membr. Sci. 2019, 586, 53-83.
- Wang, Y.; Yao, M.; Chen, Y.; Zuo, Y.; Zhang, X.; Cui, L. J. Alloys Compd. 2015, 627, 7-12.
- Zhang, Z.; Gao, P.; Cheng, J.; Liu, G.; Zhang, X.; Feng, Y. Water Res. 2018, 136, 54-63.
- Chen, Y. Y.; Ma, Y. L.; Yang, J.; Wang, L. Q.; Lv, J. M.; Ren, C. J. Chem. Eng. J. 2017, 307, 15-23.
- Urbano, B. F.; Bustamante, S.; Palacio, D. A.; Vera, M.; Rivas, B. L. Polym. Int. 2020, 69, 333-345.
- Chen, B.; Liu, Y.; Chen, S.; Zhao, X.; Meng, X.; Pan, X. J. Taiwan Inst. Chem. Eng. 2016, 67, 191-201.
- Finlay, A. C.; Hobby, G. L.; P’An, S. Y.; Regna, P. P.; Routien, J. B.; Seeley, D. B.; Shull, G. M.; Sobin, B. A.; Solomons, I. A.; Vinson, J. W.; Kane, J. H. Science. 1950, 111, 85.
- Wright, C. S.; Tschan, D. N. A. M. A. Arch. Dermatology Syphilol. 1953, 67, 125-128.
- Francke, E. L.; Neu, H. C. Med. Clin. North Am. 1987, 71, 1155-1168.
- Florez, J.; Armijo, J. A.; Mediavilla, Á. Farmacología humana Florez, 3°edicion; Manson, S. A.: Barcelona, España 1997.
- Nelson, M. L. Tetracyclines Biol. Chem. Med. 2001, 1, 3-63.
- Nelson, M. L. Adv. Dent. Res. 1998, 12, 5-11.
- Pérez-Trallero, E.; Iglesias, L. Enferm. Infecc. Microbiol. Clin. 2003, 21, 520-529.
- Klein, N. C.; Cunha, B. A. Med. Clin. North Am. 1995, 79, 789-801.
- Bel Prieto, E. Cienc. y Tecnol. Pharm. 2001, 11, 48-51.
- Oka, H.; Ikai, Y.; Kawamura, N.; Yamada, M.; Harada, K. ichi; Ito, S.; Suzuki, M. J. Agric. Food Chem. 1989, 37, 226-231.
- Loftin, K. A.; Adams, C. D.; Meyer, M. T.; Surampalli, R. J. Environ. Qual. 2008, 37, 378-386.
- Ian, C.; Marilyn, R. Microbiol. Mol. Biol. Rev. 2001, 65, 232-260.
- Dupuy, A. Farmacocinética de oxitetraciclina en dosificación oral múltiple en cerdos. Análisis PK-PD. Tesis doctoral, Universidad Complutense de Madrid: Madrid, España 2015.
- Stephens, C. R.; Murai, K.; Brunings, K. J.; Woodward, R. B. J. Am. Chem. Soc.1 956, 78, 4155-4158.
- Leeson, L. J.; Krueger, J. E.; Nash, R. A. J. New York 1963, 4155, 1155-1160.
- Qiang, Z.; Adams, C. Water Res.2004, 38, 2874-2890.
- Jones, A. D.; Bruland, G. L.; Agrawal, S. G.; Vasudevan, D. Environ. Toxicol. Chem. 2005, 24, 761-770.
- Gonzales, P.A. Estudio de depleción de Oxitetraciclina en huevos obtenidos de gallinas de postura experimentales. Tesis pregrado, Universidad de Chile, Santiago, Chile 2013.
- Tookwinas, S.; Keerativiriyaporn, S. Acquacult. Asia 2004, 9, 29-33.
- Ferreira, C. S. G.; Nunes, B. A.; Henriques-Almeida, J. M. de M.; Guilhermino, L. Ecotoxicol. Environ. Saf.2007, 67, 452-458.
- Burch, D. G. S. Examination of the pharmacokinetic/pharmacodynamic (PK/PD) relationships of orally administered antimicrobials and their correlation whit the therapy of various bacterial and mycoplasmal infections in pigs. Thesis for the Diploma of Fellowship, Royal College of Veterinary: London, England 2012.
- Agwuh, K. N.; MacGowan, A. J. Antimicrob. Chemother. 2006, 58, 256-265.
- Kolpin, D.; Furlong, E.; Zaugg, S.; Buxton, H. 2002, 36, 1999-2000.
- Rodriguez-Narvaez, O. M.; Peralta-Hernandez, J. M.; Goonetilleke, A.; Bandala, E. R. Chem. Eng. J. 2017, 323, 361-380.
- Pazda, M.; Kumirska, J.; Stepnowski, P.; Mulkiewicz, E. Sci. Total Environ.2019, 697, 134023.
- Danalıoğlu, S. T.; Bayazit, Ş. S.; Kerkez, Ö.; Alhogbi, B. G.; Abdel Salam, M. Removal of ciprofloxacin from aqueous solution using humic acid- and levulinic acid- coated Fe3O4 nanoparticles. 2017, 123, 259-267.
- Peng, X.; Hu, F.; Huang, J.; Wang, Y.; Dai, H.; Liu, Z. Microporous Mesoporous Mater.2016, 228, 196-206.
- Pan, B.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q.; Zheng, S. Chem. Eng. J.2009, 151, 19-29.
- Otero, M.; Zabkova, M.; Rodrigues, A. E. Sep. Purif. Technol. 2005, 45, 86-95.
- Lee, J. W.; Jung, H. J.; Kwak, D. H.; Chung, P. G. Water Res. 2005, 39, 617-629.
- Palacio, D. A.; Becerra, Y.; Urbano, B. F.; Rivas, B. L. Chemosphere 2020, 258, 127416.
- Tan, X.; Liu, Y.; Zeng, G.; Wang, X.; Hu, X.; Gu, Y.; Yang, Z. Chemosphere 2015, 125, 70-85.
- Reyes B, H. Rev. Med. Chil. 2020, 148, 103-108.
- Delgado, S. A.; Leyva, R. A.; Elizástegui, D. C.; Zerquera, H. A. Rev. Cuebana. Farm. 2009, 32, 164-168.
- Choi, K.J.; Kim, S. G.; Kim, C. won; Kim, S. H. Chemosphere 2007, 66, 977-984.
- Choi, K. J.; Son, H. J.; Kim, S. H. Sci. Total Environ. 2007, 387, 247-256.
- Wang, T.; Pan, X.; Ben, W.; Wang, J.; Hou, P.; Qiang, Z. J. Environ. Sci. (China) 2017, 52, 111-117.
- Yang, W.; Zheng, F.; Lu, Y.; Xue, X.; Li, N. Ind. Eng. Chem. Res.2 011, 50, 13892.
- Rivas, B.L.; Urbano, B. F.; Sánchez, J. Front. Chem. 2018, 6, 1-13.
- Rivas, B.L.; Pereira, E.D.; Palencia, M.; Sánchez, J. Prog. Polym. Sci. 2011, 36, 294-322.
- Palacio, D.A.; Rivas, B.L.; Urbano, B. F. Chem. Eng. J. 2018, 351, 85-93.
- Palacio, D.A.; Leiton, L. M.; Urbano, B.F.; Rivas, B.L. Environ. Res. 2020, 182, 109014.
- Rivas B.L,; Moreno-Villoslada I.; J. Membr. Sci. 2000, 178, 165-170.
- Geckeler K.E.; Zhou R.N.; Rivas B.L. Angew. Makromol. Chem. 1992, 197, 107-115.
- Rivas B.L.; Pooley S.A.; Luna M. Makromol. Rapid Commun. 2000, 21, 905-908.
- Rodríguez-Dorado, R.; Carro, A. M.; Chianella, I.; Karim, K.; Concheiro, A.; Lorenzo,
- R. A.; Piletsky, S.; Alvarez-Lorenzo, C. Anal. Bioanal. Chem. 2016, 408, 6845-6856.
- Lu, Y.; Lei, L.; He, J.; Zhao, T. J. Sep. Sci.2020, 43, 2172-2179.
- Aguilar, J. F. F.; Miranda, J. M.; Rodriguez, J. A.; Paez-Hernandez, M. E.; Ibarra, I. S. J. Polym. Res. 2020, 27, 1-12.

