MECHANICAL AND MORPHOLOGICAL PROPERTIES OF POLY(3-HYDROXYBUTYRATE)-THERMOPLASTIC STARCH/CLAY/EUGENOL BIONANOCOMPOSITES

- Clay, biodegradable polymers, thermoplastic starch, bionanocomposites.
Copyright (c) 2020 SChQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
Food packaging for traditional foods mainly serves to isolate foods from external environmental. For this reason, it is a growing interest in developing active packaging materials that can extend foodstuff shelf-life. Also, it could counteract the level of contamination by plastic in the environment using biodegradable polymers.
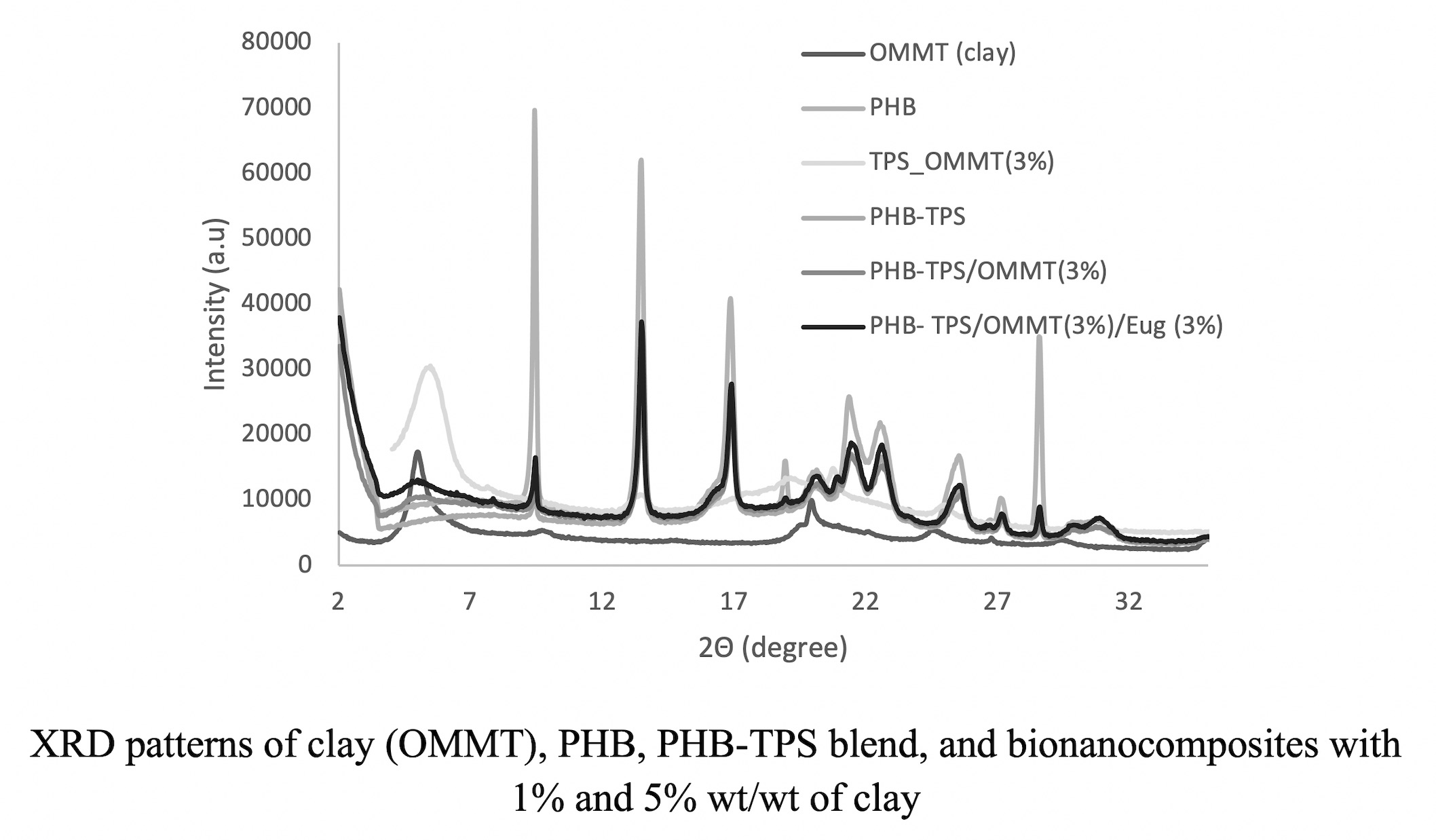
For this purpose, poly(3-hydroxybutyrate) (PHB)-thermoplastic starch (TPS)/ organically modified montmorillonite (OMMT)/eugenol bionanocomposites were prepared by melt blending. Morphology, thermal, and mechanical properties were determined by comparing the concentration influence of eugenol and clay on the PHB-TPS blend and PHB. X-ray diffraction measurements revealed the existence of exfoliated morphology in the bionanocomposites. Atomic force microscopy imaging (AFM) showed the difference in morphological characteristics of the pure PHB and the prepared bionanocomposites. Roughness analysis from the AFM data indicates that the presence of clay and TPS increases the roughness with respect to PHB. Dynamic mechanical thermal analysis (DMTA) showed that bionanocomposites have an improved storage module. Finally, properties such as hardness and elastic modulus studied by nanoindentation showed a decrease with respect to PHB. The presence of eugenol in the bionanocomposites does not show a significant effect on the different properties.
References
- A. A. Shah, F. Hasan, A. Hameed and S. Ahmed, Biotechnol. Adv., 2008, 26, 246–65.
- P. Europe, Plastics – the Facts 2019, 2019.
- P. Europe, Plastics-the facts 2017, An analysis of European plastics production, demand and waste data; 2017, 2017.
- S. Chanprateep, J. Biosci. Bioeng., 2010, 110, 621–632.
- J. Greene, J. Polym. Environ., 2007, 15, 269–273.
- S. Sinha Ray and M. Okamoto, Prog. Polym. Sci., 2003, 28, 1539–1641.
- M. M. Reddy, S. Vivekanandhan, M. Misra, S. K. Bhatia and A. K. Mohanty, Prog. Polym. Sci., 2013, 38, 1653–1689.
- Y. Dahman and C. U. Ugwu, Bioprocess Biosyst. Eng., 2014, 37, 1561–8.
- M. C. S. Corrêa, M. C. Branciforti, E. Pollet, J. a. M. Agnelli, P. a. P. Nascente and L. Avérous, J. Polym. Environ., 2011, 20, 283–290.
- G. Chen, G. Hao and T. Guo, J. Mater. Sci. Lett., 2002, 21, 1587–1589.
- A. Botana, M. Mollo, P. Eisenberg and R. M. T. Sanchez, Appl. Clay Sci. J., 2010, 47, 263–270.
- E. Panayotidou, A. Baklavaridis, I. Zuburtikudis and D. S. Achilias, J. Appl. Polym. Sci., 2015, 132, 1–8.
- D. a. D’Amico, L. B. Manfredi and V. P. Cyras, Thermochim. Acta, 2012, 544, 47–53.
- A. Giannakas, P. Stathopoulou, G. Tsiamis and C. Salmas, J. Food Process. Preserv., 2019, 1–15.
- S. Tunç and O. Duman, LWT - Food Sci. Technol., 2011, 44, 465–472.
- M. Abdollahi, M. Rezaei and G. Farzi, J. Food Eng., 2012, 111, 343–350.
- K. A. Garrido-Miranda, B. L. Rivas, M. A. Pérez -Rivera, E. A. Sanfuentes and C. Peña-Farfal, LWT - Food Sci. Technol., 2018, 98, 260–267.
- V. H. Campos-Requena, B. L. Rivas, M. A. Pérez and E. D. Pereira, Polym. Int., 2016, 65, 483–490.
- V. H. Campos-Requena, B. L. Rivas, M. A. Pérez, C. R. Figueroa, N. E. Figueroa and E. A. Sanfuentes, Postharvest Biol. Technol., 2017, 129, 29–36.
- S. Beigzadeh Ghelejlu, M. Esmaiili and H. Almasi, Int. J. Biol. Macromol., 2016, 86, 613–621.
- W. C. Oliver and G. M. Pharr, J. Mater. Res., 1992, 7, 1564–1583.
- S. Bruzaud and A. Bourmaud, Polym. Test., 2007, 26, 652–659.
- D. M. Panaitescu, C. A. Nicolae, A. N. Frone, I. Chiulan, P. O. Stanescu, C. Draghici, M. Iorga and M. Mihailescu, J. Appl. Polym. Sci., 2017, 134, 1–14.
- M. a. Abdelwahab, A. Flynn, B.-S. Chiou, S. Imam, W. Orts and E. Chiellini, Polym. Degrad. Stab., 2012, 97, 1822–1828.
- M. Zhang and N. L. Thomas, J. Appl. Polym. Sci., 2009, 116, 688–694.
- A. Arora and G. W. Padua, J. Food Sci., 2010, 75, R43-9.
- M. Farmahini-Farahani, A. Khan, P. Lu, A. H. Bedane, M. Eic and H. Xiao, Appl. Clay Sci., 2017, 135, 27–34.
- R. Hajiraissi and M. Parvinzadeh, Appl. Surf. Sci., 2011, 257, 8443–8450.
- K. A. Garrido-Miranda, B. L. Rivas and M. A. Pérez, J. Appl. Polym. Sci., 2017, 134, 45217.
- M. Parvinzadeh, S. Moradian, A. Rashidi and M.-E. Yazdanshenas, Polym. Plast. Technol. Eng., 2010, 49, 874–884.
- M. Ataeefard and S. Moradian, Appl. Surf. Sci., 2011, 257, 2320–2326.
- R. M. S. M. Thiré, T. A. A. Ribeiro and C. T. Andrade, J. Appl. Polym. Sci., 2006, 100, 4338–4347.
- L. Zaidi, S. Bruzaud, M. Kaci, A. Bourmaud, N. Gautier and Y. Grohens, Polym. Degrad. Stab., 2013, 98, 348–355.
- J. Adam, B. A. Korneliusz and M. Agnieszka, J. Appl. Polym. Sci., 2013, 130, 3175–3183.
- J.-W. Rhim, H.-M. Park and C.-S. Ha, Prog. Polym. Sci., 2013, 38, 1629–1652.
- H. M. Wilhelm, M. R. Sierakowski, G. P. Souza and F. Wypych, Carbohydr. Polym., 2003, 52, 101–110.
- F. Ivanič, D. Jochec-Mošková, I. Janigová and I. Chodák, Eur. Polym. J., 2017, 93, 843–849.
- A. M. Díez-Pascual and A. L. Díez-Vicente, ACS Appl. Mater. Interfaces, 2014, 6, 9822–34.
- Y. Q. Zhang, J. H. Lee, J. M. Rhee and K. Y. Rhee, Compos. Sci. Technol., 2004, 64, 1383–1389.

