PHYTOCHEMICAL AND CRYSTALLOGRAPHIC STUDIES OF AZARA DENTATA EXTRACTS AND ITS CYTOTOXIC EFFECTS ON HUMAN BREAST CANCER CELL, MCF-7

- Azara dentata,
- Phytochemicals,
- Antimicrobial activity,
- Cytotoxic activity,
- X-Ray Diffraction
- GC-MS. ...More
Copyright (c) 2020 SChQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
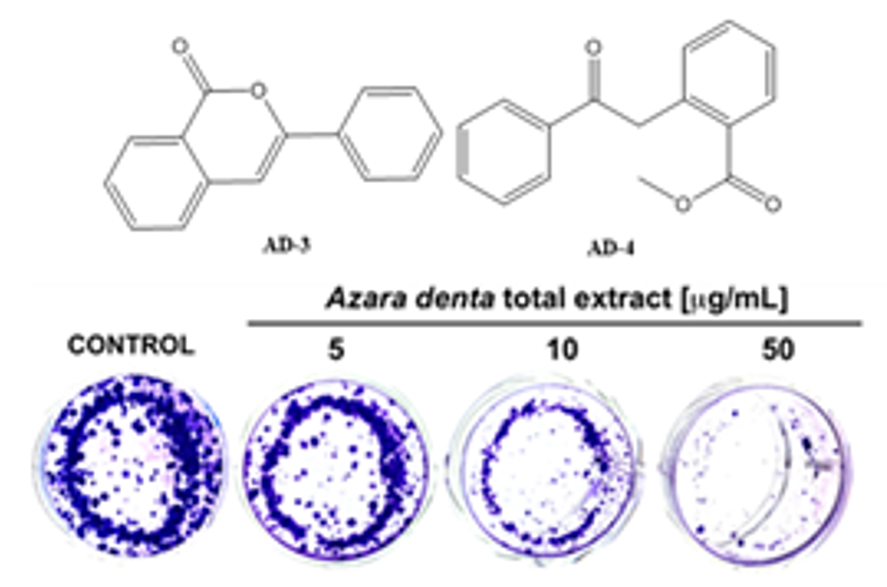
Azara dentata Ruiz & Pav. also called “Corcolen” is an endemic shrub of Chile. The honey produced in areas with abundance of Azara dentata is highly appreciate by its polyphenols. In the present work, we isolated and identified for the first time the phytochemical components of Azara dentata and its cytotoxic effects were analyzed on cancer cells together with its antimicrobial activity. The results showed that palmitic acid methyl ester, linolenic acid methyl ester, α-tocopherol and β-sitosterol are the main non-polar constituents of the plant, while the compounds AD-3 (3-phenylisocoumarin, 0.0001% yield) and AD-4 (methyl 2-phenacylbenzoate, 0.00039% yield) were isolated by column chromatography with n-hexane/EtOAc (3:2 v/v) and their structures were determined using NMR analysis. In addition, the structure of AD-4 was complemented by a single crystal x-ray structural determination. Azara dentata showed moderate antibacterial activity against S. aureus and S. epidermidis with a MIC of 5.0 and 10 mg/mL respectively. The cytotoxic activity of Azara dentata against MCF-7 cancer cells showed an IC50 of 15.63 µg/mL, this is a promissory value for the deeper study of its metabolites in cancer models.
References
- A. Giordano, M. Retamal, E. Fuentes, L. Ascar, P. Velásquez, K. Rodríguez, G. Montenegro, Food Analytical Methods, 12, 1511, (2019)
- R. Bridi, G. Montenegro. The value of chilean honey: Floral origin related to their antioxidant and antibacterial activities. Honey Analysis; Arnaut, V., Ed.; Intechopen: Rijeka, Croatia, (2017) pp 63-78.
- A. Giordano, M. Retamal, F. Leyton, P. Martínez, R. Bridi, P. Velásquez, G. Montenegro, Journal of Food. 16, 484, (2018).
- Bruker. SAINT, APEX3 and SADABS (Computer programs in the APEX3 suite). Bruker AXS Inc., Madison, Wisconsin, USA, (2015).
- G.M. Sheldrick, Acta Crystallographica, A64,112, (2008).
- G.M. Sheldrick, Acta Crystallographica, C71, 3, (2015).
- Y.N. Wang, H.X. Wang, Y.S. Jin, C.Y. Bu, J. Cheng, L.L. Zhao, G.L. Shi, Journal of economic entomology, 103, 1372, (2010).
- A. Roy, N. Thakran, N. Bharadvaja, Nat Prod Chem Res, 6, 4, (2018).
- G. A. Dhopeshwarkar, C. Subramanian, Lipids, 11, 67, (1976).
- M. Ayaz, M. Junaid, F. Ullah, F. Subhan, A. Sadiq, G. Ali, M. El-Shazly, Frontiers in pharmacology, 8, 697, (2017).
- R.G. Alscher, J.L. Hess, "Vitamin E, α-tocopherol." Antioxidants in higher plants. CRC Press, 111, 2017.
- D.W. Wu, Q.L. Liang, X.L. Zhang, Z. Jiang, X.H. Fan, W. Yue, Q. Wu, Natural Product Research, 31, 131, (2017).
- D. Lai, J. Li, S. Zhao, G. Gu, X. Gong, P. Proksch, L. Zhou, Natural Product Research, 1, 5, (2019).
- H. Zhang, H. Matsuda, A. Kumahara, Y. Ito, S. Nakamura, M. Yoshikawa, Bioorganic & medicinal chemistry letters, 17, 4972, (2007).
- K. Sudarshan, M.K. Manna, I.S. Aidhen, European Journal of Organic Chemistry, 8, 1797, (2015).
- A. Casnati, R. Maggi, G. Maestri, N. Della Ca’, E. Motti, The Journal of organic chemistry, 82, 8296, (2017).
- C.R. Groom, I.J. Bruno, M.P. Lightfoot, S.C. Ward, Acta Crystallographica, B72, 171, (2016).
- A. Gohil, S. Deshmukh, V. Bhattacharya, R. Lavhale, S. Verekar, A. Kate, Natural Product Research, 1, 9, (2019).
- H. Hussain, I.R. Green, Expert Opinion on Therapeutic Patents, 27, 1267, (2017).
- A. Saeed, European Journal of Medicinal Chemistry, 116, 290, (2016).