Co-PRECIPITATION SYNTHESIS, CHARACTERIZATION OF CoFe2O4 NANOMATERIAL AND EVALUATION OF ITS TOXICITY BEHAVIOR ON HUMAN LEUKEMIA CANCER K562 CELL LINE

- CoFe2O4,
- Nanoparticles,
- Chronic Myeloid Leukemia (CML),
- Co-precipitation,
- K562 cell
Copyright (c) 2020 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
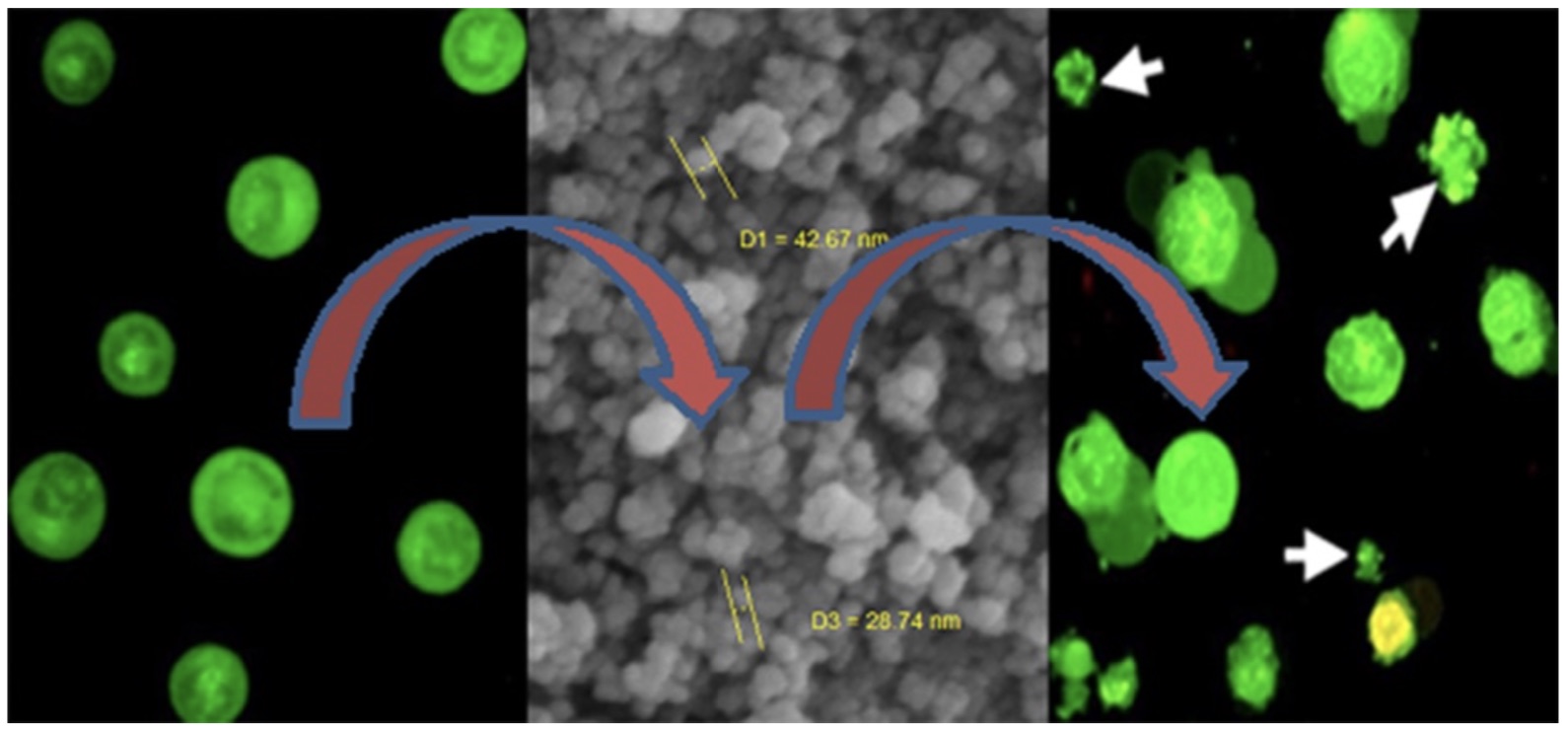
In this study, cobalt ferrite nanomaterial with inverse spinel structure was synthesized via a co-precipitation method using Co(NO3)3.6H2O and Fe(NO3)3.9H2O. A 15% NH4OH solution was used as an alkaline agent for pH adjustment. Oleic acid was used as a coating agent on the surface of the synthesized cobalt ferrite nanoparticles. PXRD analysis confirmed that the obtained material had a main CoFe2O4 cubic crystal structure with space group Fd-3m and lattice parameters a=b=c= 8.40 Å (JCPDS standard card no = 01-077-0426). According to the SEM analysis, the pseudo-spherical particles size distribution and morphology of the as-synthesized nanomaterial were homogeneous. The magnetic behavior of the obtained material was analyzed by vibrating-sample magnetometer (VSM). Besides, anti-cancer effect of the as-synthesized cobalt ferrite nanoparticles was examined on an experimental model of acute myeloid leukemia (CML) K562 cell line for the first time. Inhibition of cell growth and cytotoxicity (apoptosis) that are characteristics features of cobalt ferrite nanoparticles are tested on K562 cell line. The results showed growth and viability inhibition and induction of apoptosis in K562 cells treated with the nanoparticles.
References
- V. Kesavan, P.S. Sivanand, S. Chandrasekaran, Y. Koltypin, A. Gedanken, Angew Chem. Int. Ed. 38, 3521 (1999).
- L.H. Huo, W. Li, L.H. Lu, H.N. Cui, S. Q. Xi, J. Wang, B. Zhao, Y.C. Shen, Z.H. Lu, Chem. Mater. 12, 790 (2000).
- K. Suresh, K. C. Patil, J. Mater. Sci. Lett. 12, 572 (1993).
- M. Kroell, M. Pridoehl, G. Zimmermann, L. Pop, S. Odenbach, A. Hartwig, J. Magn. Mater. 289, 21 (2005).
- K.C. Patial, M.S. Hegde, T. Rattan, T. Aruna, Chemistry of nano crystalline oxide materials, combustion synthesis, properties and applications, Published by World Scientific Published Co. Pte. Ltd, 2008.
- J.E. Kim, J.Y. Shin, M.H. Cho, Arch. Toxicol. 86, 685 (2012).
- V.P. Torcilin, Nanoparticulates as drug carriers, 1st Edition, Imperial College Press, 2006.
- J. Ding, P.G. McCormick, Solid State Commun. 95, 313 (1995).
- K. Maaz, A. Mumtaza, S.K. Hasanain, A. Ceylan, J. Magn. Magn. Mater. 308, 289 (2007).
- A. Pui, D. Gherca, G. Carja, Dig. J. Nanomater Biostruct. 4, 1783 (2011).
- F. Liu, S. Laurent, A. Roch, L.V. Elst, R.N. Muller, J. Nanomaterials., 2013, Article ID 462540, http://dx.doi.org/10.1155/2013/462540.
- S.R. Ahmed, P. Kofinas, Macromolecules. 35, 3338 (2002).
- Y. Liu, Y. Zhang, J.D. Feng, C.F. Li, J. Shi, R. Xiong, J. Exp. Nanosc. 4, 159 (2009).
- S. Sathiya, K. Parasuraman, M. Anbarasu, K. Balamurugan, Nano Vision. 5, 133 (2015).
- F. Mansouri, G. Amiri, M. Fatemi, Nanomed J. 3, 196 (2016).
- F. Huixia, C. Baiyi, Z. Deyi, Z. Jianqiang, T. Lin, J. Magn. Magn. Mater. 35668 (2014).
- Z. Zi, Y. Sun, X. Zhu, Z. Yang, J. Dai, W. Song, J. Magn. Magn. Mater. 321, 1251 (2009).
- M. Houshiar, F. Zebhi, Z.J. Razi, A. Alidoust, Z. Askari, J. Magn. Magn. Mater. 371, 43 (2014).
- G. Sheet, A.R. Cunliffe, E.J. Offerman, C.M. Folkman, J. Appl. Phys. 107, 104309 (2010).
- V.L. Othéro de Brito, S.A. Cunha, L.V. Lemos, C.B. Nunes, Sensors. 12, 10086 (2012).
- S. Pauline, A.P. Amaliya, Arch. Appl. Sci. Res. 3, 213 (2011).
- Z. Zhou, Y. Zhang, Z. Wang, W. Wei, W. Tang, J. Shi, R. Xiong, Appl. Surf. Sci. 254, 6972 (2008).
- G.R. Amiri, S. Fatahian, A.R. Jelvani, R.M. Dehaghi, M. Habibi, Adv. Mater Rapid. Commun. 5, 1178 (2011).
- K.S. Rao, G.S.V.R.K. Choudary, K.H. Rao, C. Sujatha, Proced. Mater. Sci. 10, 19 (2015).
- W.P. Wang, H. Yang, T. Xian, J.L. Jiang, Mater Trans. 53, 1586 (2012).
- C. Sun, J.S.H. Lee, M. Zhang, Adv. Drug Deliv. Rev. 60, 1252 (2008).
- Z. Dai, R.C. Quackenbush, K.D. Courtney, M. Grove, D. Cortez, G.W. Reuther, A.M. Pendergast, Genes Dev. 12, 1415 (1998).
- R.T. Maziarz, M.J. Mauro, Bone Marrow Transplant. 32, 459 (2003).
- D. Fabbro, S. Ruetz, E. Buchdunger, S.W. Cowan-Jacob, G. Fendrich, J. Liebetanz, J. Mestan, T. O'Reilly, P. Traxler, B. Chaudhuri, H. Fretz, J. Zimmermann, T. Meyer, G. Caravatti, P. Furet, P.W. Manley, Pharm. Therap. 93, 79 (2002).
- S. Han, Y. Kim, T. Kim, BMB reports. 1 (2008).
- A. Russo, M. Terrasi, V. Agnese, D. Santini, V. Bazan, Ann. Oncol., 2006, 17, 115-123.
- H. Guo, X. Yang, T. Xiao, W. Zhang, L. Lou, J. Mugnier, Appl. Sur. Sci. 230, 215 (2004).
- G. Baldi, D. Bonacchi, M.C. Franchini, D. Gentili, G. Lorenzi, A. Ricci, C. Ravagli, Langmuir. 23, 4026 (2007).
- B. Babic-Stojic, V. Jokanovic, D. Milivojevic, Z. Jaglicic, D. Makovec, N. Jovic, M. J. Nanomater. 2013, Article ID 741036. http://dx.doi.org/10.1155/2013/741036.
- S. Hyewon, H. Oh, C. Lee, J. Microbiol Biotechnol. 152, 540 (2011).
- S. Jeong, M. Han, C. Jin, G. Kim, B. Choi, T. Nam, S. Kim, Y. Choi, Int. J. Mol. Med. 25, 31 (2010).

