EFFECT OF GERMINATION AND COOKING ON IRON CONTENT, PHYTIC ACID AND LECTINS OF FOUR VARIETIES OF CHILEAN BEANS (Phaseolus vulgaris).
- lectins,
- phytic acid,
- iron,
- Hemagglutinating
Copyright (c) 2020 SChQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
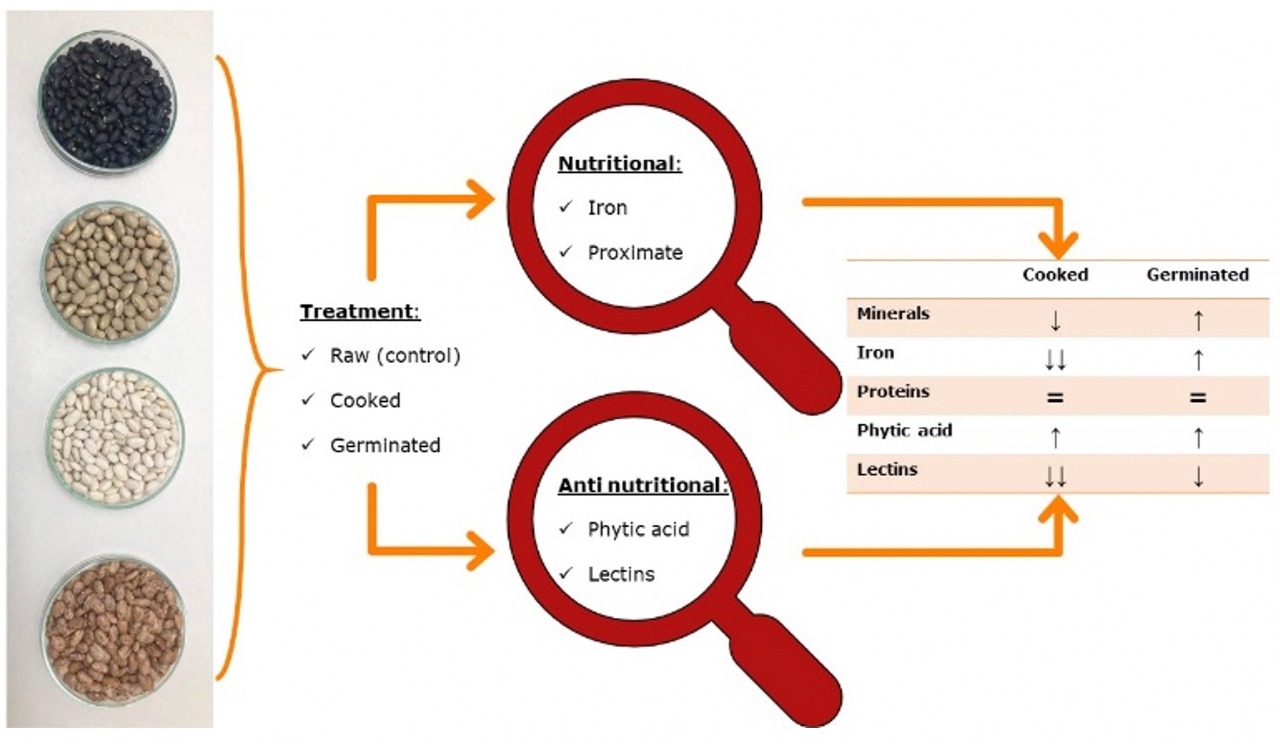
The family of Fabaceae has been fundamental into the traditional nutrition in South America, including Chile, however in this country has reduce consumption of legumes between children in school age. This investigation pursued establish the impact over nutritional and no nutritional composition of chilean beans (Phaseolus vulgaris) exposed to cook and germination. The results obtained has shown the cooked samples has decreased the contain of iron and also diminished in total minerals from ashes analysis, instead the germinated samples increased both. Total proteins determined by Kjeldahl method exhibit no change for cooked and germinated beans in relation to control without treatment, while phytic acid increased and lectins decreased with the action of cook or germination of P.vulgaris. All the analysis was supported by statistical assessment and suggest the beans germination is a good alternative to improve the variety of recipes available to prepare beans in other attractive presentations on the subject of promote the children intake.
References
- V. M. Hernández-López and M.-P. Ma. Luisa P. Vargas-Vázquez, José S. Muruaga-Martínez, Sanjuana, Hernández-Delgado, Netzahualcóyotl, “Origen, domesticación y diversificación del frijol comun. Avances y perspectivas,” Rev. Fitotec. Mex., vol. 36, no. 2, pp. 95–104, 2013.
- B. S. Gaut, “The complex domestication history of the common bean,” Nat. Genet., vol. 46, no. 7, pp. 663–664, 2014.
- V. Viviana Becerra, C. Mario Paredes, M. Carmen Rojo, L. M. Díaz, and M. W. Blair, “Microsatellite marker characterization of Chilean common bean (Phaseolus vulgaris L.) germplasm,” Crop Sci., vol. 50, no. 5, pp. 1932–1941, 2010.
- C. Baginsky G and L. Ramos C., “Situación de las legumbres en Chile: Una mirada agronómica,” Rev. Chil. Nutr., vol. 45, pp. 21–31, 2018.
- S. M. F. Bessada, J. C. M. Barreira, and M. B. P. P. Oliveira, “Trends in Food Science & Technology Pulses and food security : Dietary protein , digestibility , bioactive and functional properties,” Trends Food Sci. Technol., vol. 93, no. 228, pp. 53–68, 2019.
- F. G. B. Los, A. A. F. Zielinski, J. P. Wojeicchowski, A. Nogueira, and I. M. Demiate, “Beans (Phaseolus vulgaris L.): whole seeds with complex chemical composition,” Curr. Opin. Food Sci., vol. 19, pp. 63–71, 2018.
- T. G. Porch et al., “Nutritional composition and cooking characteristics of tepary bean (Phaseolus acutifolius Gray) in comparison with common bean (Phaseolus vulgaris L.),” Genet. Resour. Crop Evol., vol. 64, no. 5, pp. 935–953, 2017.
- A. Thakur, V. Sharma, and A. Thakur, “An overview of anti-nutritional factors in food,” Int. J. Chem. Stud., vol. 7, no. 1, pp. 2472–2479, 2019.
- N. Nciri, T. Shin, and N. Cho, “Public health hazards associated with naturally occurring toxins in legume seeds - Rapid detection and characterization of a lectin from a korean cultivated kidney bean (Phaseolus vulgaris L.),” Asian J. Pharm. Clin. Res., vol. 11, no. Special Issue 3, pp. 76–79, 2018.
- R. Shang et al., “The Diversity of Four Anti-nutritional Factors in Common Bean,” Hortic. Plant J., vol. 2, no. 2, pp. 97–104, 2016.
- J. Zhang, J. Shi, S. Ilic, S. Jun Xue, and Y. Kakuda, “Biological properties and characterization of lectin from red kidney bean (Phaseolus Vulgaris),” Food Rev. Int., vol. 25, no. 1, pp. 12–27, 2009.
- L. U. Thompson, “Potential health benefits and problems associated with antinutrients in foods,” Food Res. Int., vol. 26, no. 2, pp. 131–149, 1993.
- A. Pinheiro, C. Ivanovic, and L. Rodríguez, “Consumo de legumbres en Chile. perspectivas y desafíos,” Rev. Chil. Nutr., vol. 45, pp. 14–20, 2018.
- M. J. Barros Cruz, “El poroto como metáfora de la nación, los sujetos populares y la revolución en ‘Rotología del Poroto’ de Pablo de Rokha,” Rev. Chil. Lit., no. 90, pp. 55–78, 2015.
- I. M. Aguilera Bornard, “Historia e identidad: elementos para pensar el consumo de porotos en Chile,” Interciencia, vol. 43, no. May, pp. 379–384, 2018.
- S. Suzanne Neilsen, Food Analysis Laboratory Manual, Third Edit. Springer, 2017.
- Y. Gao et al., “A modified colorimetric method for phytic acid analysis in soybean,” Crop Sci., vol. 47, no. 5, pp. 1797–1803, 2007.
- L. Shi, S. D. Arntfield, and M. Nickerson, “Changes in levels of phytic acid, lectins and oxalates during soaking and cooking of Canadian pulses,” Food Res. Int., vol. 107, no. March, pp. 660–668, 2018.
- H. M. Sánchez-Arteaga, J. E. Urías-Silvas, H. Espinosa-Andrews, and E. García-Márquez, “Effect of chemical composition and thermal properties on the cooking quality of common beans (Phaseolus vulgaris),” CYTA - J. Food, vol. 13, no. 3, pp. 385–391, 2015.
- B. Venkidasamy, D. Selvaraj, A. S. Nile, S. Ramalingam, G. Kai, and S. H. Nile, “Indian pulses: A review on nutritional, functional and biochemical properties with future perspectives,” Trends Food Sci. Technol., vol. 88, no. March, pp. 228–242, 2019.
- M. Olivares, F. Pizarro, S. De Pablo, M. Araya, and R. Uauy, “Iron, Zinc, and Copper: Contents in Common Chilean Foods and Daily Intakes in Santiago, Chile,” Nutrition, vol. 20, no. 2, pp. 205–212, 2004.
- V. Kumar, A. Rani, S. Rajpal, G. Srivastava, A. Ramesh, and O. P. Joshi, “Phytic acid in Indian soybean: Genotypic variability and influence of growing location,” J. Sci. Food Agric., vol. 85, no. 9, pp. 1523–1526, 2005.
- P. Y. Lin and H. M. Lai, “Bioactive compounds in legumes and their germinated products,” J. Agric. Food Chem., vol. 54, no. 11, pp. 3807–3814, 2006.
- L. X. López-Martínez, N. Leyva-López, E. P. Gutiérrez-Grijalva, and J. B. Heredia, “Effect of cooking and germination on bioactive compounds in pulses and their health benefits,” J. Funct. Foods, vol. 38, pp. 624–634, 2017.
- R. A. Habiba, “Changes in anti-nutrients, protein solubility, digestibility, and HCl-extractability of ash and phosphorus in vegetable peas as affected by cooking methods,” Food Chem., vol. 77, no. 2, pp. 187–192, 2002.
- X. Guo, T. Li, K. Tang, and R. H. Liu, “Effect of germination on phytochemical profiles and antioxidant activity of mung bean sprouts (Vigna radiata),” J. Agric. Food Chem., vol. 60, no. 44, pp. 11050–11055, 2012.
- F. Manan, T. Hussain, I. Alli, and P. Iqbal, “Effect of cooking on phytic acid content and nutritive value of Pakistani peas and lentils,” Food Chem., vol. 23, no. 2, pp. 81–87, 1987.
- E. A. Shimelis and S. K. Rakshit, “Effect of processing on antinutrients and in vitro protein digestibility of kidney bean (Phaseolus vulgaris L.) varieties grown in East Africa,” Food Chem., vol. 103, no. 1, pp. 161–172, 2007.
- E. Doria, B. Campion, F. Sparvoli, A. Tava, and E. Nielsen, “Anti-nutrient components and metabolites with health implications in seeds of 10 common bean (Phaseolus vulgaris L. and Phaseolus lunatus L.) landraces cultivated in southern Italy,” J. Food Compos. Anal., vol. 26, no. 1–2, pp. 72–80, 2012.
- A. K. Ramírez-Jiménez, R. Reynoso-Camacho, M. E. Tejero, F. León-Galván, and G. Loarca-Piña, “Potential role of bioactive compounds of Phaseolus vulgaris L. on lipid-lowering mechanisms,” Food Res. Int., vol. 76, no. P1, pp. 92–104, 2015.
- K. MuraMoto, “Lectins as bioactive proteins in foods and feeds,” Food Sci. Technol. Res., vol. 23, no. 4, pp. 487–494, 2017.


