µ-BENZENE-1,2,4,5-TETRACARBOXYLATO-κ2O1:O4)BIS[ AQUABIS- 2-(AMINOMETHYL) PYRIDINE-κ2N,N') NICKEL (II)] DECAHYDRATE: STRUCTURE AND MAGNETIC PROPERTIES
- crystal structure,
- nickel complex,
- magnetism
Copyright (c) 2019 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
µ-Benzene-1,2,4,5-tetracarboxylato-κ2O1:O4)bis[aquabis-2-(aminomethyl) pyridine-κ2N,N') nickel (II)] decahydrate, [Ni2(amp)4(btc)(H2O)2].10H2O has been synthesized and its crystal structure determined by X-ray diffraction. The complex crystallizes in the Monoclinic space group P21/n with cell dimensions a= 11.0102 (10) Å, b= 21.5416 (19) Å, c= 11.0445 (10) Å and ß= 118.753 (1)°, and two formula units per cell (Z= 2, Z’= 0.5).
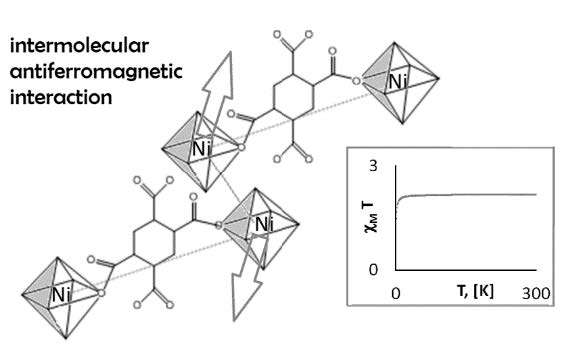
The structure consists of centrosymmetric dimeric units in which the benzene tetracarboxylate anions (btc) bridge the symmetry–related nickel (II) cations, two bidentate chelating molecules of 2-(aminomethyl) pyridine and a terminal aqua ligand completing the six-fold coordination at each metal. These dimers link to each other via H-bonding to form chains parallel to [100], while the water solvates form a strongly bound 2D network parallel to (100), made up by small (R44(8) ) and large (R20 20(40)) rings, the latter ones “pierced” by the chains of dimmers, with a profuse H-bonding interconnection between both substructures.
Magnetic susceptibility measurements as a function of temperature disclose weak interdimeric exchange interactions. Fitting of these data to a dimeric model yields J= -3.5 cm-1 and g= 2.13.
References
- L. Carneiro, A. R. Silva, P. S. Shuttleworth, V. Budarin and J. H.Clark, Molecules, 19 (8), pp 11988-11998 (2014), doi.org/10.3390/molecules190811988.
- G H. Le Bozec, T. Renouard, Eur. J. Inorg. Chem., pp 229-239 (2000), doi.org/10.1002/(SICI)1099-0682(200002)2000:2<229::AIDEJIC229>3.0.CO;2-A.
- Ch-Ch. Wang, Y-Q. Zhang, T. Zhu, X-Y. Zhang, S-J. Gao, Polyhedron, 90, pp 58-68 (2015), doi.org/10.1016/j.poly.2015.01.042; A.M. Aref , A. I. El-Said, R. Gabr, R. A. Mukred., American Chemical Science Journal, 6(3), pp 144-157( 2015), doi.10.9734/ACSJ/2015/15754
- K. L. Haas and K. J. Franz, Chem Rev., 109 (10), pp 4921–4960 (2009), doi: 10.1021/cr900134a; Ch-Ti Chen, K. S.Suslick., Coordination Chemistry Reviews. 128, pp 293-322 (1993), doi.org/10.1016/0010-8545(93)80036-5
- D.Q Chu, J.Q. Xu, L. M., Wang, A.Q.Tang,l.Ye, Eur. J. Inorg. Chem., pp 1135–1137. (2001), doi.org/10.1002/1099-0682(200105)2001:5<1135::AIDEJIC1135>3.0.CO;2-G; Y. Wu, C. Xia, J. Qian , J. Xie, J. Chem. Sci. 129 (8), pp 1183–1191(2017), doi 10.1007/s12039-017-1319-x
- Y. B Go, X Wang, EV Anokhina, A.J. Jacobson, Inorg. Chem., 44 , pp 8265–8271 (2005), doi: 10.1021/ic050644d
- F. Rochon Fernande, G.Massarweh, Inorg.Chim. Acta, 304, pp 190–198 (2000), doi.org/10.1016/S0020-1693(00)00086-4
- A.M. Atria, M. T.Garland, R. Baggio, Acta Cryst., C70, pp 541–546 (2014), doi:10.1107/S2053229614008134
- R.J. Doedens, Prog. Inorg. Chem., 21, pp 209-231 (1976), doi.org/10.1002/9780470166222.ch9; Y-Z. Zheng, Z. Zhenga, X-M. Chen, Coordination Chemistry Reviews, 258–259 pp 1–15(2014), doi: 10.1016/j.ccr.2013.08.031
- M. Kurmoo., Chem. Soc. Rev., 38 pp 1353-1379 (2009), doi:10.1039/B804757J: N. R. de Campos, M. A. Ribeiro, W. X. C. Oliveira, D. O. Reis,H. O. Stumpf, A.C., Doriguetto, F. C. Machado,C. B. Pinheiro, F. Lloret, M. Julve, J. Cano, M. V. Marinho, Dalton Trans., 45, pp172-189 (2016), doi:10.1039/c5dt03401a
- S. Sen, M.K Saha, T. Gupta, A.K. Karmakar, P. Kundu, S. Mitra, M.B. Hursthouse, K. M. A. Malik, Journal of Chemical Crystallography, 28, pp 771–777 (1998), doi.org/10.1023/A:102182072
- E. Colacio, J. M. Domínguez-Vera, M. Ghazi, R. Kivekäs, M. Klinga, J. M.Moreno, Eur. J. Inorg. Chem. pp 441- 445 (1999), doi.org/10.1002/(SICI)1099-0682(199903)1999:3<441::AIDEJIC441>3.0.CO;2-T
- Bruker SMART, V5.624. Data Collection Software. Siemens Analytical X-ray, Instruments Inc., Madison, Wisconsin, USA. (2001).
- Bruker (SAINT, V6.22A (Including SADABS). Data Reduction Software.Siemens., Analytical X-ray Instruments Inc., Madison, Wisconsin, USA. (2002).
- G.M. Sheldrick, Acta Cryst., A64, pp 112–122 (2008), doi.org/10.1107/S0108767307043930
- G. M Sheldrick, Acta Cryst., C71, pp 3–8 (2015), doi.org/10.1107/S2053229614024218
- Earnshaw, Introduction to magnetochemistry, Academic Press, London (1968).
- G. De Munno, T. Poerio, M. Julve, F. Lloret, A. Derory, J. Chem. Soc. Dalton Trans., pp 1179-1184 (1993), doi:10.1039/DT9930001179
- M. J. Prushan, D. M. Tomezsko, S. Lofland, M. Zeller, A. D. Hunter, Inorg. Chim. Acta, 360 pp 2245–2254(2007), doi.org/10.1016/j.ica.2006.11.008
- Y.-T. Li, C.- W. Yan, Ch.- S. Xu, D.- Z. Liao., Synth. React. Inorg. Met. Org. Chem., 28:3, pp 367-381(1998), doi.org/10.1080/00945719809349361
- H.-L. Zhu, Y.-X. Tong, X.-M. Chen, Ch.-X. Ren., Trans. Metal Chem., 26, pp. 528-531 (2001), doi.org/10.1023/A:1011011422776
- E.-Q. Gao, Q.-H. Zhao, J.-K. Tang, D.-Z. Liao, Z.-H. Jiang, S.-P. Yan, J. Coord. Chem., 55:2, pp 205-213 (2002), doi.org/10.1080/00958970211878
- K. S. Bürger, P. Chaudhuri, K. Wieghardt, B. Nuber, Chem. Eur. J. 1pp. 583-593 (1995), doi.org/10.1002/chem.19950010904


