OPTIMIZATION AND VALIDATION OF A LIQUID CHROMATOGRAPHIC METHOD FOR DETERMINATION OF CAPSAICIN IN CHILI PEPPERS
- Capsaicinoids,
- capsaicin,
- fluorescence,
- mass spectrometry,
- chromatography
Copyright (c) 2019 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
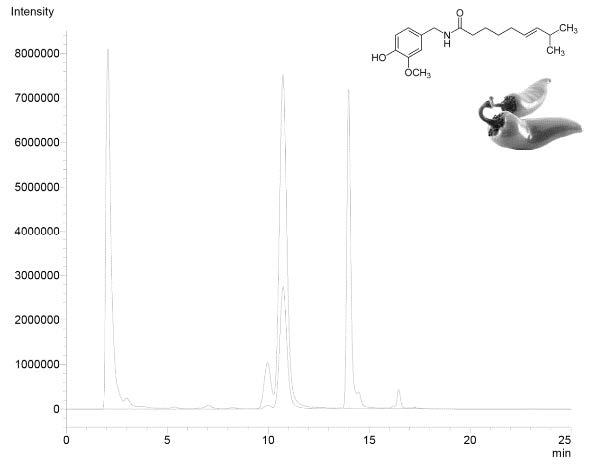
Capsaicinoids are organic compounds present in several foods like chili peppers. This group of molecules are responsible of fruit pungency, as well as, of several healthy properties. The present work reports an optimized and validated liquid chromatography method coupled to fluorescence and mass spectrometry detectors for a selective determination of capsaicin in Chili peppers (Capsicum annum and C. pendulum). To the best our knowledge this is the first report about capsaicin determination in chili peppers commercialized in Chile. Chromatographic conditions were optimized giving the following optimal conditions: 76% organic phase (MeOH: ACN: acetic acid (50:50:0.1 v/v/v) at 9 min of gradient program and column temperature of 35°C. With these conditions capsaicin, dihydrocapsaicin and nordihydrocapsaicin were separated in 15 min. Data calibration curve (0.01-2.00 mg L-1) fitted a linear regression model with a determination coefficient (R2) of 0.9986. Repeatability (relative standard deviation, RSD) and intermediate precision (RSD) showed values of 1.51% (n=6) and 1.04% (n=3), respectively. Recovery (n=3) at three levels ranged from 94.80 to 109.40%, (RDS <2.39%). The method was applied to analyze 10 chili peppers varieties commercialized in Chile. A broad range of capsaicin and dihydrocapsaicin contents were observed, finding values from 0.1 up to 127.3 μg g-1.
References
- World Health Organization (WHO), Obesity and overweight. (2018) acces date: February 21, 2018.
- A. Stein, E. Rodríguez-Cerezo, Functional food in the European Union. Joint Research Centre. European Commission. (2008).
- I. Perucka, M. Materska, Innov Food Sci Emerg 2, 189, (2001).
- A. Topuz, F. Ozdemir, J Food Compost Anal 20, 596, (2007).
- M. Sganzerla, J.P. Coutinho, A.M.T. de Melo, H.T. Godoy, Food Res Int 64, 718, (2014).
- A. Morales-Soto, A.M. Gómez-Caravaca, P. García-Salas, A. Segura-Carretero, A. Fernández-Gutiérrez, Food Res Int 51, 977, (2013).
- G.F. Barbero, M. Palma, C.G. Barroso, Anal Chim Acta 578, 227, (2006).
- M.F. Sadayoshi Kosuge, Agr Biol Chem 34, 248, (1970).
- R.L. Jarret, B. Perkins, T. Fan, A. Prince, K. Guthrie, B. Skoczenski, J Food Compost Ana 16, 189, (2003).
- M.G. Usman, M.Y. Rafii, M.R. Ismail, M.A. Malek, M.A. Latif, Molecules 19, 6474, (2014).
- E. Morales-Soriano, B. Kebede, R. Ugás, T. Grauwet, A. Van Loey, M. Hendrickx, Food Res Int 109, 250, (2018).
- A. Schmidt, G. Fiechter, E.-M. Fritz, H.K. Mayer, J Food Compost Ana 60, 32, (2017).
- M.M. Backonja, Eur J Pain 4, 170, (2010).
- E. Becerra-Martínez, E. Florentino-Ramos, N. Pérez-Hernández, L. Gerardo Zepeda-Vallejo, N. Villa-Ruano, M. Velázquez-Ponce, F. García-Mendoza, A.E. Bañuelos-Hernández, Food Res Int 102, 163, (2017).
- L. Lopez-Carrillo, M. Hernandez Avila, R. Dubrow, Am J Epidemiol 139, 263, (1994).
- V.S. Govindarajan, U.J. Salzer, Crit Rev Food Sci 23, 207, (1986).
- E.C.H.C. Protection, Opinion of the Scientific Committee on Food on Capsaicin. (2002).
- W.L. Scoville., J Am Pharm Assoc 1, 453, (1912).
- A.M. Krajewska, J.J. Powers, J Chromatogr A 409, 223, (1987).
- O. Monago-Maraña, M. Guzmán-Becerra, A. Muñoz de la Peña, T. Galeano-Díaz, J Food Compost Anal 67, 10, (2018).
- P.K.S. Bhuvaneshwari S, Alice Kuruvilla, Int. j. pharm. biol. sci. arch 4, (2013).
- W.-K. Ryu, H.-W. Kim, G.-D. Kim, H.-I. Rhee, J Food Drug Anal 25, 798, (2017).
- G.F. Barbero, A. Liazid, M. Palma, C.G. Barroso, Talanta 75, 1332, (2008).
- T. Bajer, P. Bajerová, D. Kremr, A. Eisner, K. Ventura, J Food Compost Anal 40, 32, (2015).
- M.D. Collins, L.M. Wasmund, P.W. Bosland, HortScience 30, 137, (1995).
- P.H. Todd, M.G. Bensinger, T. Biftu, J Food Sci 42, 660, (1977).
- S.L.C. Ferreira, R.E. Bruns, E.G.P. da Silva, W.N.L. dos Santos, C.M. Quintella, J.M. David, J.B. de Andrade, M.C. Breitkreitz, I.C.S.F. Jardim, B.B. Neto, J. Chromatogr. A 1158, 2, (2007).
- I.C.o.H. (ICH), 2005.
- R.I.S. Jörgen Vessman, Jacobus F. van Staden, Klaus Danzer, Wolfgang Lindner, Duncan Thorburn Burns, Aleš Fajgelj, Helmut Müller, Pure Appl. Chem 73, 1381, (2001).
- O. Cisneros-Pineda, L.W. Torres-Tapia, L.C. Gutiérrez-Pacheco, F. Contreras-Martín, T. González-Estrada, S.R. Peraza-Sánchez, Food Chem 104, 1755, (2007).
- A. Peña-Alvarez, L.A. Alvarado, L.E. Vera-Avila, Instrum Sci. Technol. 40, 429, (2012).
- T. Stipcovich, G.F. Barbero, M. Ferreiro-González, M. Palma, C.G. Barroso, Food Chem 239, 217, (2018).
- K.G. Sweat, J. Broatch, C. Borror, K. Hagan, T.M. Cahill, Food Chem 210, 606, (2016).
- Z.A.A. Othman, Y.B.H. Ahmed, M.A. Habila, A.A. Ghafar, Molecules 16, 8919, (2011).
- G.F. Barbero, A.G. Ruiz, A. Liazid, M. Palma, J.C. Vera, C.G. Barroso, Food chem 153, 200, (2014).
- L. Duelund, O.G. Mouritsen, Food Chem 221, 913, (2017).


