CONFOCAL MICROSCOPY STUDIES OF LIVING FUNGAL HYPHAE AND CONIDIA USING RHENIUM (I) TRICARBONYL COMPLEXES AS FLUORESCENT DYES
Copyright (c) 2019 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
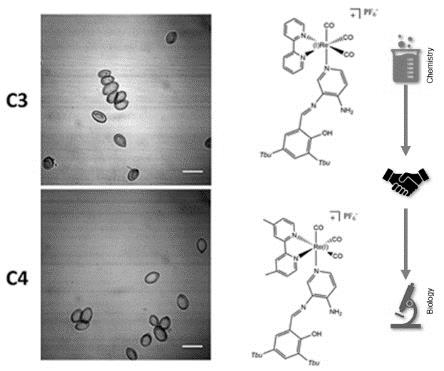
Botrytis cinerea is a ubiquitous necrotrophic filamentous fungal pathogen, causing the “grey mold” disease in a wide range of plants, including species of importance in the food industry. Recently, we reported the use of fac-[Re(I)(CO)3(2,2’-bpy)L]PF6 (C3) and fac-[Re(I)(CO)3(4,4’-dimethyl-2,2’-bpy)L]PF6 (C4) complexes as biomarkers for yeasts and bacteria. Nevertheless, the use of these complexes to stain filamentous fungi, such as Botrytis cinerea, has not been explored so far. Since Botrytis cinerea exhibits a dynamic multilayer cell wall that changes the composition during normal growth, in both their hyphae and conidia, is not possible to directly extrapolate the use of both C3 and C4 as biomarkers for Botrytis cinerea without an experimental approach. In this work, we explored new features of the luminescent compounds C3 and C4 as biomarkers for the higher filamentous fungi Botrytis cinerea, including conidia and juvenile hyphae. We found that, with the new protocol proposed, both C3 and C4 were suitable to stain conidia with a simple procedure since no cell permeabilization is required. Additionally, these results suggest that C3 and C4 can selectively stain living conidia, allowing the differentiation from non-germinating conidia. This point opens a new focus of development for rhenium (I) tricarbonyl complexes as new fluorescent biomarkers for Botrytis cinerea with potential properties as vital staining.
References
- Williamson, B., et al., Botrytis cinerea: the cause of grey mould disease. Mol Plant Pathol, 2007. 8(5): p. 561-80.
- Tripathi, P., N.K. Dubey, and A.K. Shukla, Use of some essential oils as post-harvest botanical fungicides in the management of grey mould of grapes caused by Botrytis cinerea. World Journal of Microbiology and Biotechnology, 2007. 24(1): p. 39-46.
- Gonçalves, M.R. and K.P.M. Frin, Synthesis, characterization, photophysical and electrochemical properties of rhenium(I) tricarbonyl diimine complexes with triphenylphosphine ligand. Polyhedron, 2017. 132: p. 20-27.
- Mukuta, T., et al., Photochemical Processes in a Rhenium(I) Tricarbonyl NHeterocyclic Carbene Complex Studied by Time-Resolved Measurements. Inorg Chem, 2017. 56(6): p. 3404-3413.
- Machura, B., et al., Tricarbonyl rhenium(I) complex of benzothiazole–Synthesis, spectroscopic characterization, X-ray crystal structure and DFT calculations. Journal of Organometallic Chemistry, 2013. 724: p. 82-87.
- Sacksteder, L., et al., Luminescence Studies of Pyridine Alpha-Diimine Rhenium(I) Tricarbonyl Complexes. Inorg Chem, 1990. 29(21): p. 4335-4340.
- Kia, R. and F. Safari, Synthesis, spectral and structural characterization and computational studies of rhenium(I)-tricarbonyl nitrito complexes of 2,2′- bipyridine and 2,9-dimethylphenanthroline ligands: π-Accepting carácter of the diimine ligands. Inorganica Chimica Acta, 2016. 453: p. 357-368.
- Kaplanis, M., et al., Re(I) tricarbonyl complex of 1,10-phenanthroline-5,6-dione: DNA binding, cytotoxicity, anti-inflammatory and anti-coagulant effects towards platelet activating factor. J Inorg Biochem, 2014. 135: p. 1-9.
- Thorp-Greenwood, F.L., An Introduction to Organometallic Complexes in Fluorescence Cell Imaging: Current Applications and Future Prospects. Organometallics, 2012. 31(16): p. 5686-5692.
- Raszeja, L.J., et al., Asymmetric rhenium tricarbonyl complexes show superior luminescence properties in live cell imaging. Chem Commun (Camb), 2017. 53(5): p. 905-908.
- Thorp-Greenwood, F.L., et al., Tris(rhenium fac-tricarbonyl) Polypyridine Functionalized Cyclotriguaiacylene Ligands with Rich and Varied Emission. Organometallics, 2016. 35(11): p. 1632-1642.
- Carreno, A., et al., Fluorescence probes for prokaryotic and eukaryotic cells using Re(CO)(3)(+) complexes with an electron withdrawing ancillary ligand. New Journal of Chemistry, 2016. 40(9): p. 7687-7700.
- Carreño, A., et al., X-ray diffraction and relativistic DFT studies on the molecular biomarker fac-Re(CO)3(4,4′-dimethyl-2,2′-bpy)(E-2-((3-aminopyridin-4-ylimino)-methyl)-4,6-di-tert-butylphenol)(PF6). Chemical Papers, 2017. 71(10): p. 2011-2022.
- Carreño, A., et al., Spectral, theoretical characterization and antifungal properties of two phenol derivative Schiff bases with an intramolecular hydrogen bond. New J. Chem., 2015. 39(10): p. 7822-7831.
- Cantu, D., et al., Characterization of the cell wall of the ubiquitous plant pathogen Botrytis cinerea. Mycol Res, 2009. 113(Pt 12): p. 1396-403.
- Buckley, P.M., V.E. Sjaholm, and N.F. Sommer, Electron microscopy of Botrytis cinerea conidia. J Bacteriol, 1966. 91(5): p. 2037-44.
- Hawker, L.E. and R.J. Hendy, An Electron-Microscope Study of Germination of Conidia of Botrytis cinerea. J Gen Microbiol, 1963. 33: p.43-6.
- Carreno, A., et al., Experimental and theoretical studies of the ancillary ligand (E)-2-((3-amino-pyridin-4-ylimino)-methyl)-4,6-di-tert-butylphenol in the rhenium(I) core. New Journal of Chemistry, 2015. 39(7): p. 5725-5734.
- Carreno, A., et al., Spectral, theoretical characterization and antifungal properties of two phenol derivative Schiff bases with an intramolecular hydrogen bond. New Journal of Chemistry, 2015. 39(10): p. 7822-7831.
- Herzog, W., et al., Electron transfer between hydrogen-bonded pyridylphenols and a photoexcited rhenium(I) complex. Chemphyschem, 2013. 14(6): p. 1168-76.
- Wenger, O.S., Proton-coupled electron transfer with photoexcited metal complexes. Acc Chem Res, 2013. 46(7): p. 1517-26.
- Carreño, A., et al., Substituted bidentate and ancillary ligands modulate the bioimaging properties of the classical Re(i) tricarbonyl core with yeasts and bacteria. New J. Chem., 2017. 41(5): p. 2140-2147.
- Leuschner, R.G. and P.J. Lillford, Investigation of bacterial spore structure by high resolution solid-state nuclear magnetic resonance spectroscopy and transmission electron microscopy. Int J Food Microbiol, 2001. 63(1-2): p.35-50.
- Hussey, M.A. and A. Zayaitz, Endospore stain protocol. American Society of Microbiology, 2007.
- Gull, K. and A.P.J. Trinci, Fine Structure of Spore Germination in Botrytis cinerea. Journal of General Microbiology, 1971. 68(2): p. 207-220.


